Saturday's live enrichment session with Sunburst (March 2, 2013)
Saturday's live enrichment session with Carson (March 2, 2013)
Thursday's frozen enrichment session with Carson (Jan. 31, 2013)
Thursday's frozen enrichment session with Carson (Jan. 24, 2013)
Hatchling Buddy Swim (mid-November, 2012)
Hatchlings getting used to environment (mid-November, 2012)
Hatchling snapshot before aggressive behavior

Harold, another loggerhead hatchling at 3 months old, about to aggressively bite the medium-sized woffle ball containing shrimp.
Hatchling snapshot during high interaction

Rooney, loggerhead hatchling (at 3 months old), interacting with small-sized woffle ball containing shrimp. Since this session we have changed to medium-sized woffle balls since those are easier for the hatchlings ,at their current stage of development, to access.
"Feeding Enrichment in an Opportunistic Carnivore: The Red Fox" Abstract
Kistler, Claudia, Daniel Hegglin, Hanno Wurbel, and Barbra Konig. "Feeding Enrichment in an Opportunistic Carnivore: The Red Fox." Applied Animal Behaviour Science 116.2-4 (2009): 260-65. 2009. Web. 3 Jan. 2013.
This is a reliable source since Kistler is associated with a university in Switzerland, Hegglin is associated with an urban and wildlife research facility, while Konig is associated with a German university. The article begins by stressing the need for enrichment for captive animals. The authors then discuss the importance of captive animals exhibiting foraging behaviors. Furthermore, “feeding enrichment based on the animals’ natural foraging strategies may be crucial for the wellbeing of captive carnivores (Lindburg, 1998; Bashaw et al., 2003)” (261). Foxes are opportunistic carnivores who constantly search for food, making dietary enrichment extremely beneficial to captive foxes. Several studies are cited comparing dietary enrichment between carnivorous species. The researchers hypothesized that the most amount of activity would be exhibited by the fox when the dietary enrichment was placed randomly within its enclosure (at a random time). In order to examine this idea, the researchers used four different feeding methods: electronic feeders alone or “in combination with (ii) a self-service food box (unpredictable in time plus time-consuming manipulation), (iii) scattered and hidden food (unpredictable in time and space plus time-consuming), or (iv) an electronic dispenser
(highly unpredictable in time and space plus time consuming). We predicted that general activity and behavioural diversity would increase from treatment (i) through treatment (iv)” (261). The authors describe the enclosure as well as the foxes. In order to assess behavioral diversity, the researchers created an ethogram based on another study. Over the course of the study, all food was placed within the various feeding devices thirty minutes prior to observations.
In the end, the researchers discovered that through dietary enrichment, behavioral diversity amongst the foxes increased. Differentiating between the five different feeding techniques used, the first one had the lowest activity. Similar to the hypothesis, activity was at it’s peak when the food was unpredictable in time and space. Again the authors note that all four foxes exhibited increased activity with all four feeding techniques as well as temporal and spatial ignorance also heightened activity. At the end, the authors suggest further research by examining whether there is a relationship between increased activity and behavioral diversity (through feeding techniques) improve an animal’s well being. Overall I thought the data, statistical analysis, and graphs were difficult to understand.
This is a reliable source since Kistler is associated with a university in Switzerland, Hegglin is associated with an urban and wildlife research facility, while Konig is associated with a German university. The article begins by stressing the need for enrichment for captive animals. The authors then discuss the importance of captive animals exhibiting foraging behaviors. Furthermore, “feeding enrichment based on the animals’ natural foraging strategies may be crucial for the wellbeing of captive carnivores (Lindburg, 1998; Bashaw et al., 2003)” (261). Foxes are opportunistic carnivores who constantly search for food, making dietary enrichment extremely beneficial to captive foxes. Several studies are cited comparing dietary enrichment between carnivorous species. The researchers hypothesized that the most amount of activity would be exhibited by the fox when the dietary enrichment was placed randomly within its enclosure (at a random time). In order to examine this idea, the researchers used four different feeding methods: electronic feeders alone or “in combination with (ii) a self-service food box (unpredictable in time plus time-consuming manipulation), (iii) scattered and hidden food (unpredictable in time and space plus time-consuming), or (iv) an electronic dispenser
(highly unpredictable in time and space plus time consuming). We predicted that general activity and behavioural diversity would increase from treatment (i) through treatment (iv)” (261). The authors describe the enclosure as well as the foxes. In order to assess behavioral diversity, the researchers created an ethogram based on another study. Over the course of the study, all food was placed within the various feeding devices thirty minutes prior to observations.
In the end, the researchers discovered that through dietary enrichment, behavioral diversity amongst the foxes increased. Differentiating between the five different feeding techniques used, the first one had the lowest activity. Similar to the hypothesis, activity was at it’s peak when the food was unpredictable in time and space. Again the authors note that all four foxes exhibited increased activity with all four feeding techniques as well as temporal and spatial ignorance also heightened activity. At the end, the authors suggest further research by examining whether there is a relationship between increased activity and behavioral diversity (through feeding techniques) improve an animal’s well being. Overall I thought the data, statistical analysis, and graphs were difficult to understand.
"Happy Herpetofauna at Melbourne Zoo!" Abstract
Hobbs, Raelene. "Happy Herpetofauna at Melbourne Zoo!" 38-41 The Royal Melbourne Zoological Gardens, 2 Nov. 2006. Web. 3 Jan. 2013.
This is a reliable journal article since the author is associated with the Melbourne Zoo. The article begins by discussing the zoo’s reptile house’s history and the inhabitants (at the time this article was written). The diversity of reptiles is then explained. The author notes that enclosure size and design are key with enriching a reptile; larger exhibits allow maximum room for interaction with multiple stimuli. The Melbourne staff changes the appearance of the exhibits on a regular basis in order to encourage investigative behavior(s). After describing substrates and various perches for lizards and geckos, environmental enrichment is finally discussed. Similar to the sea turtles at the Pittsburgh Zoo & PPG Aquarium, various reptiles at Melbourne experience natural basking during the warmer months since UVA/B is an important factor for an ectotherm’s life. Swimming and olfactory enrichment are then discussed, followed by dietary enrichment. Similar to my study where we observe the turtles’ behavior with live food, Melbourne occasionally feeds goldfish to their crocodiles for physical stimulation. Lighting, water quality, and correct temperatures are key, the author notes, to successful enrichment with reptiles. Overall this article was extremely basic, therefore I was disappointed at the lack of depth provided during the discussions of environmental and dietary enrichment.
This is a reliable journal article since the author is associated with the Melbourne Zoo. The article begins by discussing the zoo’s reptile house’s history and the inhabitants (at the time this article was written). The diversity of reptiles is then explained. The author notes that enclosure size and design are key with enriching a reptile; larger exhibits allow maximum room for interaction with multiple stimuli. The Melbourne staff changes the appearance of the exhibits on a regular basis in order to encourage investigative behavior(s). After describing substrates and various perches for lizards and geckos, environmental enrichment is finally discussed. Similar to the sea turtles at the Pittsburgh Zoo & PPG Aquarium, various reptiles at Melbourne experience natural basking during the warmer months since UVA/B is an important factor for an ectotherm’s life. Swimming and olfactory enrichment are then discussed, followed by dietary enrichment. Similar to my study where we observe the turtles’ behavior with live food, Melbourne occasionally feeds goldfish to their crocodiles for physical stimulation. Lighting, water quality, and correct temperatures are key, the author notes, to successful enrichment with reptiles. Overall this article was extremely basic, therefore I was disappointed at the lack of depth provided during the discussions of environmental and dietary enrichment.
"Effectiveness of environmental enrichment in reducing stereotypic behaviour in captive Australian Sea Lions (Neophoca cinerea)" Abstract
Smith, Bradley, and Carla Litchfield. "Effectiveness of Environmental Enrichment in Reducing Stereotypic Behaviour in Captive Australian Sea Lions (Neophoca Cinerea)." 23-37. The Royal Melbourne Zoological Gardens, 2 Nov. 2006. Web. 3 Jan. 2013.
This journal article is a credible source since both authors are associated with the University of South Australia. The article begins by describing the life of a captive sea lion as well as marine mammals in general. Through citation of another study, the authors notes that captivity causes negative effects particularly on sea lions. The authors note that due to the presence of novel objects, sea lions perform exploratory behavior, increase play behavior and social interactions; these objects are encouraged to be used for goal-directed behavior in order to limit stereotypic behaviors. There is limited research on the success of the varying types of enrichment with sea lions, no research on Australian Sea Lions (Neophoca cinerea). In addition to this, many zoological institutions do not have the available resources to conduct behavioral studies with animal interactions with enrichment. The varying types enrichment used by sea lions include “natural enrichment items (e.g., driftwood, kelp, exhibits with large pools and beaches), artificial enrichment items (e.g., balls, food treat balls, Frisbees) & training (see The Shape of Enrichment for further ideas on enrichment with seals and sea lions)” (24). The authors then discuss what previous studies have shown about decreases of stereotypic behaviors seen in sea lions and the studies’ limitations. This study aims to further the scientific knowledge of captivity and sea lions as well as become a guide to zoological institutions with seals and sea lions to further their enrichment programs in addition to better husbandry protocol.
The sea lions of this study were observed six hours each day over a thirty day period, “incorporating all significant events during a typical day (e.g., feeding time), in order to get an accurate picture of sea lion behaviour in captivity at Adelaide Zoo” (26). Observations were noted during consecutive days in order to establish behavior patterns, trends, and changes in behavior. Whether the enrichments reduce the occurrence of stereotypical behaviors was also examined during this study. The authors then describe the sea lions’ living conditions as well as their background stories.
Due to no research on Australian Sea Lions available, the authors created their own ethogram based on ethograms used in previous studies of other sea lion species. The first table describes typical Australian Sea Lion behavior while the second table categorizes all of the previously described behaviors into three categories: active, inactive, or stereotypic. The authors then describe the difference between intrinsic reinforcement and extrinsic reinforcement; “Intrinsic reinforcement occurs when simply performing the behaviour increases the probability that it will occur again (Tarou & Bashaw, in press). Extrinsic reinforcement occurs when the performance of behaviour results in a consequence that is external to the behaviour itself and therefore increases the likelihood that the behaviour will recur (Tarou & Bashaw, in press)” (28-9). They then differentiate extinction and habituation; “[e]xtinction occurs when reinforcement is no longer provided for a behaviour which results in a decrease in the performance of that behaviour” (29) while habituation is when activity is decreased to repeated stimulation. The authors then describe a couple of the enrichment devices used during the study, followed by a table describing the experimental design and the total hours of observation.
Each session was two hours long, occurring three times each day, in order to examine behaviors over the course of the day (i.e during feeding time vs. placing sea lions into enclosure for the evening). Behavior and location were noted every two minutes during these sessions, totalling to 180 hours of collected observations. The authors then note that over the course of this study, zoo management did not introduce any new enrichment devices to the sea lions. In addition to this, feeding schedule was not altered. The differences between the sea lions and their eating habits are discussed. The authors then transition to statistical analysis.
Within the baseline the male exhibited pattern swimming the majority of the time, compared to the female where there was a low percentage of pattern swimming. With the beginning of enrichment interaction, more active behavior was observed with the male while it’s stereotypical behaviors decreased through interaction with food and non-food enrichment; “Although more time was spent interacting with the non-food related item, the food related item was more effective in reducing pattern swimming” (32). Unlike the male the female did not exhibit stereotypical behavior(s), suggesting that the enrichment devices alone caused an increase of active behavior (32). In addition to this, the female did not use the food enrichment compared to using the non-food enrichment. Since the study was conducted over consecutive days, habituation was present. The authors then transition to the discussion.
Again, they note that the male exhibited stereotypical behavior while the female did not while both types of enrichment lead to positive behavior(s). The occurrence of pattern swimming was decreased in the male while both sea lions exhibited an increase in active behaviors. The authors then discuss the differences in behavior between the sea lions, based on gender, by citing a couple of sources. It is then suggested that the behavior between captive male and female sea lions needs to be further investigated. The authors note that the pattern swimming of the male generally occurred in the same area of the pool at the same time of day, lasting up to one and a half hours. The authors support this idea through citing a couple of studies. They suggest to use non-food items in future studies since it is a cost effective, low maintenance enrichment method that can also inhibit interactions between sea lions. The researchers suggest further research using “more enrichment devices, items and techniques must be designed and their effectiveness tested, in order to provide enough items to choose from” (35). The authors conclude with a series of questions pertaining to either enrichment in general or enrichment for sea lions. The major issue I had with this article is that the results of the study were not present.
This journal article is a credible source since both authors are associated with the University of South Australia. The article begins by describing the life of a captive sea lion as well as marine mammals in general. Through citation of another study, the authors notes that captivity causes negative effects particularly on sea lions. The authors note that due to the presence of novel objects, sea lions perform exploratory behavior, increase play behavior and social interactions; these objects are encouraged to be used for goal-directed behavior in order to limit stereotypic behaviors. There is limited research on the success of the varying types of enrichment with sea lions, no research on Australian Sea Lions (Neophoca cinerea). In addition to this, many zoological institutions do not have the available resources to conduct behavioral studies with animal interactions with enrichment. The varying types enrichment used by sea lions include “natural enrichment items (e.g., driftwood, kelp, exhibits with large pools and beaches), artificial enrichment items (e.g., balls, food treat balls, Frisbees) & training (see The Shape of Enrichment for further ideas on enrichment with seals and sea lions)” (24). The authors then discuss what previous studies have shown about decreases of stereotypic behaviors seen in sea lions and the studies’ limitations. This study aims to further the scientific knowledge of captivity and sea lions as well as become a guide to zoological institutions with seals and sea lions to further their enrichment programs in addition to better husbandry protocol.
The sea lions of this study were observed six hours each day over a thirty day period, “incorporating all significant events during a typical day (e.g., feeding time), in order to get an accurate picture of sea lion behaviour in captivity at Adelaide Zoo” (26). Observations were noted during consecutive days in order to establish behavior patterns, trends, and changes in behavior. Whether the enrichments reduce the occurrence of stereotypical behaviors was also examined during this study. The authors then describe the sea lions’ living conditions as well as their background stories.
Due to no research on Australian Sea Lions available, the authors created their own ethogram based on ethograms used in previous studies of other sea lion species. The first table describes typical Australian Sea Lion behavior while the second table categorizes all of the previously described behaviors into three categories: active, inactive, or stereotypic. The authors then describe the difference between intrinsic reinforcement and extrinsic reinforcement; “Intrinsic reinforcement occurs when simply performing the behaviour increases the probability that it will occur again (Tarou & Bashaw, in press). Extrinsic reinforcement occurs when the performance of behaviour results in a consequence that is external to the behaviour itself and therefore increases the likelihood that the behaviour will recur (Tarou & Bashaw, in press)” (28-9). They then differentiate extinction and habituation; “[e]xtinction occurs when reinforcement is no longer provided for a behaviour which results in a decrease in the performance of that behaviour” (29) while habituation is when activity is decreased to repeated stimulation. The authors then describe a couple of the enrichment devices used during the study, followed by a table describing the experimental design and the total hours of observation.
Each session was two hours long, occurring three times each day, in order to examine behaviors over the course of the day (i.e during feeding time vs. placing sea lions into enclosure for the evening). Behavior and location were noted every two minutes during these sessions, totalling to 180 hours of collected observations. The authors then note that over the course of this study, zoo management did not introduce any new enrichment devices to the sea lions. In addition to this, feeding schedule was not altered. The differences between the sea lions and their eating habits are discussed. The authors then transition to statistical analysis.
Within the baseline the male exhibited pattern swimming the majority of the time, compared to the female where there was a low percentage of pattern swimming. With the beginning of enrichment interaction, more active behavior was observed with the male while it’s stereotypical behaviors decreased through interaction with food and non-food enrichment; “Although more time was spent interacting with the non-food related item, the food related item was more effective in reducing pattern swimming” (32). Unlike the male the female did not exhibit stereotypical behavior(s), suggesting that the enrichment devices alone caused an increase of active behavior (32). In addition to this, the female did not use the food enrichment compared to using the non-food enrichment. Since the study was conducted over consecutive days, habituation was present. The authors then transition to the discussion.
Again, they note that the male exhibited stereotypical behavior while the female did not while both types of enrichment lead to positive behavior(s). The occurrence of pattern swimming was decreased in the male while both sea lions exhibited an increase in active behaviors. The authors then discuss the differences in behavior between the sea lions, based on gender, by citing a couple of sources. It is then suggested that the behavior between captive male and female sea lions needs to be further investigated. The authors note that the pattern swimming of the male generally occurred in the same area of the pool at the same time of day, lasting up to one and a half hours. The authors support this idea through citing a couple of studies. They suggest to use non-food items in future studies since it is a cost effective, low maintenance enrichment method that can also inhibit interactions between sea lions. The researchers suggest further research using “more enrichment devices, items and techniques must be designed and their effectiveness tested, in order to provide enough items to choose from” (35). The authors conclude with a series of questions pertaining to either enrichment in general or enrichment for sea lions. The major issue I had with this article is that the results of the study were not present.
“Environmental Enrichment and Development of Cage Stereotypy in Orange-winged Amazon Parrots (Amazona Amazonica)” Abstract
Meehan, C. L., J. P. Garner, and J. A. Mench. "Environmental Enrichment and Development of Cage Stereotypy in Orange-winged Amazon Parrots (Amazona Amazonica)." Developmental Psychobiology 44.4 (2004): 209-18. Print.
This is a credible journal article since all of the authors are affiliated with a university in California. The authors begin by defining what stereotypic behaviors are as seen within captive animals. They then explain various ways in which these stereotypic behaviors can be decreased. For this study the team used Orange-winged Amazon Parrots (Amazona Amazonica), which tend to have long lives.When housed in poor conditions, they develop oral and locomotive stereotypic behaviors; “between 5 and 85% of their active time performing stereotypies (Meehan, 2002)”(210). Foraging behaviors of avian species is limited through oral stereotypic behavior, demonstrated within other studies. In addition to this, “the development of locomotor stereotypies is related to lack of space and physical complexity (Keiper, 1969)” (210). Since this avian species in particular spends a significant amount of time in the wild demonstrating foraging behaviors, this study focuses on the “introduction of foraging opportunity and physical complexity designed to increase locomotor activity” (210) since foraging and locomotive behaviors are restricted within a captive setting.
During the 48 week period the young parrots were either placed within a controlled, sterile cage or in a cage where locomotive and foraging opportunities were implemented. Through these differing environments, the authors were able to examine whether the presence of these activities reduced or ended the occurrence of the stereotypic behaviors. The authors then describe the cage dimensions and its contents as well as the parrots’ diet. The study used foraging and physical enrichment. Physical enrichment forced parrots to either “chew through barriers, manipulate objects through holes, sort through inedible material, or open containers to obtain food items” (211). During the study, twenty-four different enrichments (twelve physical, twelve foraging) were observed while eight different enrichments (four physical, four foraging) were given during each of the three 16-week periods. In order to eliminate observer interference with the parrots during observations, all examinations were recorded via videotape. The authors then discuss the specific time frame during data collection. A table is then displayed describing each of the enrichments used over the course of this study.
The authors mention that due to limited research conducted examining stereotypical behaviors in this avian species, the researchers created their own ethogram describing each of the behaviors that were observed. The ethogram was created after watching hundreds of hours of video footage from a pilot study. To monitor the birds performance, their behavior was recorded two hours each day (once in the morning, another in the afternoon); where the footage was later analyzed through a specific data collection software from Microsoft. During this time, durations of “general locomotion, preening, feeding/drinking and enrichment use, locomotor stereotypy, and oral stereotypy” (212) were noted while the amount of activity time (excluding resting and sleeping) was calculated from each tape. The authors then discuss how the data was statistically analyzed.
In the end the researchers determined that it generally took the parrot between one and twenty minutes after placement (of the enrichment) to approach them followed by investigation. Usually the parrots used the physical enrichment in order to approach the foraging enrichment. The authors then explain the graphs through discussion of various percentages of both the control group and the enriched group. Based on figure one and three, it is clear that both groups had a higher percentage of active time with the foraging enrichment compared to active time spent in the presence of physical enrichment.
Within the discussion the authors mention that their hypothesis was correct- parrots in the presence of foraging and physical enrichment spent less time performing stereotypical behaviors, compared to those within the control group. They also note that the control group performed both oral and locomotive stereotypical behavior while the enriched group only displayed oral stereotypical behavior, suggesting the the development of stereotypic behaviors is due to the lack of specific environmental objects (216). In other words, foraging enrichment can eliminate oral stereotypical behaviors since the parrot is being stimulated compared to being frustrated and stressed within a sterile, un-enriched environment. Furthermore, small cage size might develop locomotive stereotypical behavior due to inability to fly. However each stereotypical behavior cannot be assessed separately since the researchers presented physical and foraging enrichment simultaneously. The authors then discuss the length of the delay of the onset of stereotypical behaviors. Followed by a discussion of controlled enrichment as well as development of stereotypic behaviors. At the end, the researchers suggest further investigation with “patterns of stereotypy reversal” across different species.
Overall this study was somewhat difficult to comprehend mainly due to the graphs and statistical analysis. However both tables as well as figures one and three were easy to interpret.
This is a credible journal article since all of the authors are affiliated with a university in California. The authors begin by defining what stereotypic behaviors are as seen within captive animals. They then explain various ways in which these stereotypic behaviors can be decreased. For this study the team used Orange-winged Amazon Parrots (Amazona Amazonica), which tend to have long lives.When housed in poor conditions, they develop oral and locomotive stereotypic behaviors; “between 5 and 85% of their active time performing stereotypies (Meehan, 2002)”(210). Foraging behaviors of avian species is limited through oral stereotypic behavior, demonstrated within other studies. In addition to this, “the development of locomotor stereotypies is related to lack of space and physical complexity (Keiper, 1969)” (210). Since this avian species in particular spends a significant amount of time in the wild demonstrating foraging behaviors, this study focuses on the “introduction of foraging opportunity and physical complexity designed to increase locomotor activity” (210) since foraging and locomotive behaviors are restricted within a captive setting.
During the 48 week period the young parrots were either placed within a controlled, sterile cage or in a cage where locomotive and foraging opportunities were implemented. Through these differing environments, the authors were able to examine whether the presence of these activities reduced or ended the occurrence of the stereotypic behaviors. The authors then describe the cage dimensions and its contents as well as the parrots’ diet. The study used foraging and physical enrichment. Physical enrichment forced parrots to either “chew through barriers, manipulate objects through holes, sort through inedible material, or open containers to obtain food items” (211). During the study, twenty-four different enrichments (twelve physical, twelve foraging) were observed while eight different enrichments (four physical, four foraging) were given during each of the three 16-week periods. In order to eliminate observer interference with the parrots during observations, all examinations were recorded via videotape. The authors then discuss the specific time frame during data collection. A table is then displayed describing each of the enrichments used over the course of this study.
The authors mention that due to limited research conducted examining stereotypical behaviors in this avian species, the researchers created their own ethogram describing each of the behaviors that were observed. The ethogram was created after watching hundreds of hours of video footage from a pilot study. To monitor the birds performance, their behavior was recorded two hours each day (once in the morning, another in the afternoon); where the footage was later analyzed through a specific data collection software from Microsoft. During this time, durations of “general locomotion, preening, feeding/drinking and enrichment use, locomotor stereotypy, and oral stereotypy” (212) were noted while the amount of activity time (excluding resting and sleeping) was calculated from each tape. The authors then discuss how the data was statistically analyzed.
In the end the researchers determined that it generally took the parrot between one and twenty minutes after placement (of the enrichment) to approach them followed by investigation. Usually the parrots used the physical enrichment in order to approach the foraging enrichment. The authors then explain the graphs through discussion of various percentages of both the control group and the enriched group. Based on figure one and three, it is clear that both groups had a higher percentage of active time with the foraging enrichment compared to active time spent in the presence of physical enrichment.
Within the discussion the authors mention that their hypothesis was correct- parrots in the presence of foraging and physical enrichment spent less time performing stereotypical behaviors, compared to those within the control group. They also note that the control group performed both oral and locomotive stereotypical behavior while the enriched group only displayed oral stereotypical behavior, suggesting the the development of stereotypic behaviors is due to the lack of specific environmental objects (216). In other words, foraging enrichment can eliminate oral stereotypical behaviors since the parrot is being stimulated compared to being frustrated and stressed within a sterile, un-enriched environment. Furthermore, small cage size might develop locomotive stereotypical behavior due to inability to fly. However each stereotypical behavior cannot be assessed separately since the researchers presented physical and foraging enrichment simultaneously. The authors then discuss the length of the delay of the onset of stereotypical behaviors. Followed by a discussion of controlled enrichment as well as development of stereotypic behaviors. At the end, the researchers suggest further investigation with “patterns of stereotypy reversal” across different species.
Overall this study was somewhat difficult to comprehend mainly due to the graphs and statistical analysis. However both tables as well as figures one and three were easy to interpret.
"The Effect of Environmental Enrichment on the Foraging Behavior of Garter Snakes - Thamnophis Sirtalis" Abstract
Puskar, Ramona. "The Effect of Environmental Enrichment on the Foraging Behavior of Garter Snakes - Thamnophis Sirtalis." Summer 2009. Web. 11 Dec. 2012. <http://faculty.mckendree.edu/ scholars/summer2009/puskar.htm>.
Puskar begins the journal article by defining environmental enrichment followed by an example. This study investigates environmental enrichment with garter snakes. After defining environmental enrichment and giving another example, the author discusses how temperature plays a role during garter snake foraging behavior(s) since they tend to be more active within warmer environments. Description of a study with environmental enrichment and mammals is given followed by noting that there is limited research on reptiles and environmental enrichment. Puskar then hypothesizes that the common garter snake will find and capture their prey faster within an enriched environment compared to an unenriched one; therefore enrichment does play a role within foraging behavior(s) of common garter snakes. Eight young common garter snakes (sharing size and age, all bred and raised in captivity) were chosen for this study. Three subspecies of the common garter snake were also selected for this study. Four females and four males were examined, placing two of each gender into each group, which were split up based on subspecies. The author then describes the tanks where each snake inhabited. The snakes were fed either one large or two small minnows once a week on test days. The fish were place at the end of an obstacle course within a water bowl. Each time the snake was tested, the fish was placed in a different place inside the aquarium. After each trial all of the tank’s contents were removed and cleaned ( in order to remove all odors) while the time to get the fish was recorded during a two and a half hour period. If it took the snake longer amount of time, the snake was left in the aquarium in order to eat the fish. In the end environmental enrichment does not play a role within foraging behavior(s) with garter snakes. The analysis was difficult to understand due to a lack of understanding about t tests. If this study is repeated, Puskar suggests to limit human interference. The author then reminds the reader that temperature plays a significant role within a snake’s life; “On warm days the snakes would have been more willing to eat a large prey item while on cooler days they might not have desired to eat”. A difficulty that the author experienced was controlling the temperature for the snakes. Other factors that can affect a snake’s foraging behavior includes shedding cycles (controlled by growth and hormones), individual temperament, and general health. The amount of shedding depends on the snake’s size and gender; for example a young male snake sheds more frequently than an adult male snake since it grows at a much faster rate. The author notes how young snakes are not as habituated to human handling as older snakes. During this experiment two snakes died (possibly due to vitamin deficiency through a restricted diet or bacteria/parasitic infection). While this experiment did not support the author’s hypothesis, they did learn more about foraging behavior of garter snakes. Besides the data analysis this journal article was easy to comprehend and analyze.
Puskar begins the journal article by defining environmental enrichment followed by an example. This study investigates environmental enrichment with garter snakes. After defining environmental enrichment and giving another example, the author discusses how temperature plays a role during garter snake foraging behavior(s) since they tend to be more active within warmer environments. Description of a study with environmental enrichment and mammals is given followed by noting that there is limited research on reptiles and environmental enrichment. Puskar then hypothesizes that the common garter snake will find and capture their prey faster within an enriched environment compared to an unenriched one; therefore enrichment does play a role within foraging behavior(s) of common garter snakes. Eight young common garter snakes (sharing size and age, all bred and raised in captivity) were chosen for this study. Three subspecies of the common garter snake were also selected for this study. Four females and four males were examined, placing two of each gender into each group, which were split up based on subspecies. The author then describes the tanks where each snake inhabited. The snakes were fed either one large or two small minnows once a week on test days. The fish were place at the end of an obstacle course within a water bowl. Each time the snake was tested, the fish was placed in a different place inside the aquarium. After each trial all of the tank’s contents were removed and cleaned ( in order to remove all odors) while the time to get the fish was recorded during a two and a half hour period. If it took the snake longer amount of time, the snake was left in the aquarium in order to eat the fish. In the end environmental enrichment does not play a role within foraging behavior(s) with garter snakes. The analysis was difficult to understand due to a lack of understanding about t tests. If this study is repeated, Puskar suggests to limit human interference. The author then reminds the reader that temperature plays a significant role within a snake’s life; “On warm days the snakes would have been more willing to eat a large prey item while on cooler days they might not have desired to eat”. A difficulty that the author experienced was controlling the temperature for the snakes. Other factors that can affect a snake’s foraging behavior includes shedding cycles (controlled by growth and hormones), individual temperament, and general health. The amount of shedding depends on the snake’s size and gender; for example a young male snake sheds more frequently than an adult male snake since it grows at a much faster rate. The author notes how young snakes are not as habituated to human handling as older snakes. During this experiment two snakes died (possibly due to vitamin deficiency through a restricted diet or bacteria/parasitic infection). While this experiment did not support the author’s hypothesis, they did learn more about foraging behavior of garter snakes. Besides the data analysis this journal article was easy to comprehend and analyze.
The Biology of Sea Turtles vol. 1 Chapter 8 Abstract
Bjorndal, Karen A. "Chapter 8: Foraging Ecology and Nutrition of Sea Turtles." Biology of Sea Turtles. Vol. 1. 199-204, 210-13, 218-21.: CRC-Press, 1997. 199-206. Web. 10 Dec. 2012. <http://home.eckerd.edu/~meylanpa/BI200/Meylan_Web_Site/pdfs%20of%20readiings/14-Bjorndal_1997_foraging.pdf>.
This chapter begins by stating that all organisms eat based on their environment. Thisfood gives the organism nutrients, allowing it to grow leading to sexual maturity. Therefore the environment plays a role on a species’ population and conservation efforts. Through citing another source, the author claims that there are “differences in mean growth rate of green sea turtles from different foraging areas of the Hawaiian Archipelago” (200). She then suggests that by “understanding how much nutrition regulates productivity [we can] move to the next level to address how we can enhance or modify productivity to improve the survival outlook of these endangered species” (200). The chapter then transitions into informing the reader that the conclusions of the following studies discussed may be misinterpreted due to a) small sample size, b) analysis of fecal samples, c) not reporting reproductive status, and/or d) turtles tend to eat a different diet during mating and nesting season compared to other times of the year. The author then discusses green sea turtles.
Green sea turtles often live in open ocean environments. Through citation of other studies, green sea turtles are mainly carnivorous during their hatchling stage. Apparently their plasma (liquid part of blood) changes color based on the turtles diet; “after they have begun eating on sea grasses, their plasma has yellow pigmentation” (201). The author then briefly mentions that green sea turtles are found in Australia, Hawaii, Indian Ocean, Western Pacific, and Arabian Sea. The reader is informed that around a carapace (shell) of 20 to 25cm, the diet turns to herbivore (primarily sea grasses, reefs, and algae) while occassionaly eating jellyfish, salps, and sponges. The author then discusses in great detail the percentages of different species of algae found in various green sea turtles stomachs across the globe. She then discusses loggerhead sea turtles.
Loggerhead sea turtles begin their lives in shallow, open ocean environments, usually in the Atlantic. However they are also found in Australia, West Florida Shelf, South Africa, southeastern US, and the Gulf of Mexico. Similar to the discussion of green sea turtles, various contents of post-hatchling and juvenile loggerheads’ stomachs as well as fecal samples are discussed in great detail, demonstrating the variety of their omnivore diet. Towards the end of the page the author mentions that loggerheads tend to forage on the bottom of their environment in order to eat hard-shelled prey. After describing five other species of sea turtles, the author discusses diet selection.
While diet selection of sea turtles as a whole is unknown, scientists are aware that it is possibly at the hatchling stage food preference can be created as well as “reversed” (210). The author then notes that a green sea turtle’s diet can either be based on preference or “by the relative abundance of different diet species” (210). Food preference could be due to “green turtle visually scan[ing] the potential foraging area while slowly moving among algae patches” (210). In other words, this quotation (citing a study) proves that greens have selective foraging behavior. Through citation of another study, the author makes the reader note that greens never mix algae with seagrass(es) when they are both present within the same environment. In addition to this notion, the digestive tracts of greens who eat algae looks very different to greens who eat seagrass(es) since seagrass gets more digested than algae based on their structural carbohydrates varying a lot. In other words, the microbes within a green’s digestive tract will be either favored towards algae or seagrass(es) based on food preference determined at hatchling stage (211). Then the content gets confusing since it starts to talk about mixed diets being beneficial for greens due to limited plant life in some environments as well as maximizing energy levels by digesting a mixed diet even though by citing a couple of studies, the diet can not be mixed. The author then transitions into the role of sea turtles in structuring foraging habitat communities.
In the beginning of this section, the author notes that if there’s a high population of sea turtles in one area, they can be damaging to the ecosystem by being “major predators and grazers” (212). The section then discusses how greens help with nutrient cycling of various species of algae. The author then subtly hints at further research of quantifying grazing on algae by green sea turtles on coral reefs and how it “must have significant effects on percent cover by algae in these ecosystems” (212). At the end of the page the loggerhead is mentioned as a damaging predator by decreasing the “mortality factor for saucer scallops (Amusium japonicum balloti) around Bundaberg, Australia and for large juvenile and adult queen conch (Strombus gigas) in the Bahamas” (212). More research is suggested in terms of investigating competition between sea turtles and other species or compared to other sea turtles. But then the author mentions that since different species have varying foraging methods, competition amongst food resources is minimized. For example Kemp’s Ridley and Loggerheads are both located around Long Island, NY as well as Virginia (213). The author also mentions that, when in the same environment, some herbivore species of fish specifically chose to eat different algae species compared to green sea turtles in order to eliminate competition over food resources. After describing digestion, which is not needed for this study, the author transitions into Anthropogenic effects on foraging ecology and nutrition with a specific focus on marine debris.
Young sea turtles are constantly threatened by floating debris. Percentages of post-hatchling loggerheads ingesting tar and plastic demonstrate how this is a key factor against conservation efforts. More facts are mentioned, making the reader note that small amounts of debris can kill a sea turtle. Despite “mortality resulting from ingestion of debris” being difficult to estimate and assess, the damage that debris causes to sea turtles has been studied and discussed time and time again. The author notes that the “[e]ffect of nutrient dilution on intake is being being evaluated in loggerhead hatchlings” (219). The next section discusses commercial fisheries.
Right of the bat, incidental capture of sea turtles is another damaging factor to their increasing mortality rates. Fishing degrades foraging habitats, specifically trawling can destroy whole communities by decreasing the quantity and quality. Another idea for further study is investigating competition of food resources between humans and sea turtles. Through commercial fishing more and more species of fish are becoming commercially extinct leading to disruptions in food webs (due to population shifts and habitat degradation) as well as decreasing food rates of sea turtles. Furthermore sea grasses grow slowly or not at all within areas covered in scars from anchors. Oil spills also threatened sea turtle species as well as their food distribution.
In closing, the author suggests further research between relationships between foraging ecology and sea turtles. This study needs to involve wild sea turtles and natural diet(s). Overall this chapter was extremely easy to comprehend and analyze for this abstract.
This chapter begins by stating that all organisms eat based on their environment. Thisfood gives the organism nutrients, allowing it to grow leading to sexual maturity. Therefore the environment plays a role on a species’ population and conservation efforts. Through citing another source, the author claims that there are “differences in mean growth rate of green sea turtles from different foraging areas of the Hawaiian Archipelago” (200). She then suggests that by “understanding how much nutrition regulates productivity [we can] move to the next level to address how we can enhance or modify productivity to improve the survival outlook of these endangered species” (200). The chapter then transitions into informing the reader that the conclusions of the following studies discussed may be misinterpreted due to a) small sample size, b) analysis of fecal samples, c) not reporting reproductive status, and/or d) turtles tend to eat a different diet during mating and nesting season compared to other times of the year. The author then discusses green sea turtles.
Green sea turtles often live in open ocean environments. Through citation of other studies, green sea turtles are mainly carnivorous during their hatchling stage. Apparently their plasma (liquid part of blood) changes color based on the turtles diet; “after they have begun eating on sea grasses, their plasma has yellow pigmentation” (201). The author then briefly mentions that green sea turtles are found in Australia, Hawaii, Indian Ocean, Western Pacific, and Arabian Sea. The reader is informed that around a carapace (shell) of 20 to 25cm, the diet turns to herbivore (primarily sea grasses, reefs, and algae) while occassionaly eating jellyfish, salps, and sponges. The author then discusses in great detail the percentages of different species of algae found in various green sea turtles stomachs across the globe. She then discusses loggerhead sea turtles.
Loggerhead sea turtles begin their lives in shallow, open ocean environments, usually in the Atlantic. However they are also found in Australia, West Florida Shelf, South Africa, southeastern US, and the Gulf of Mexico. Similar to the discussion of green sea turtles, various contents of post-hatchling and juvenile loggerheads’ stomachs as well as fecal samples are discussed in great detail, demonstrating the variety of their omnivore diet. Towards the end of the page the author mentions that loggerheads tend to forage on the bottom of their environment in order to eat hard-shelled prey. After describing five other species of sea turtles, the author discusses diet selection.
While diet selection of sea turtles as a whole is unknown, scientists are aware that it is possibly at the hatchling stage food preference can be created as well as “reversed” (210). The author then notes that a green sea turtle’s diet can either be based on preference or “by the relative abundance of different diet species” (210). Food preference could be due to “green turtle visually scan[ing] the potential foraging area while slowly moving among algae patches” (210). In other words, this quotation (citing a study) proves that greens have selective foraging behavior. Through citation of another study, the author makes the reader note that greens never mix algae with seagrass(es) when they are both present within the same environment. In addition to this notion, the digestive tracts of greens who eat algae looks very different to greens who eat seagrass(es) since seagrass gets more digested than algae based on their structural carbohydrates varying a lot. In other words, the microbes within a green’s digestive tract will be either favored towards algae or seagrass(es) based on food preference determined at hatchling stage (211). Then the content gets confusing since it starts to talk about mixed diets being beneficial for greens due to limited plant life in some environments as well as maximizing energy levels by digesting a mixed diet even though by citing a couple of studies, the diet can not be mixed. The author then transitions into the role of sea turtles in structuring foraging habitat communities.
In the beginning of this section, the author notes that if there’s a high population of sea turtles in one area, they can be damaging to the ecosystem by being “major predators and grazers” (212). The section then discusses how greens help with nutrient cycling of various species of algae. The author then subtly hints at further research of quantifying grazing on algae by green sea turtles on coral reefs and how it “must have significant effects on percent cover by algae in these ecosystems” (212). At the end of the page the loggerhead is mentioned as a damaging predator by decreasing the “mortality factor for saucer scallops (Amusium japonicum balloti) around Bundaberg, Australia and for large juvenile and adult queen conch (Strombus gigas) in the Bahamas” (212). More research is suggested in terms of investigating competition between sea turtles and other species or compared to other sea turtles. But then the author mentions that since different species have varying foraging methods, competition amongst food resources is minimized. For example Kemp’s Ridley and Loggerheads are both located around Long Island, NY as well as Virginia (213). The author also mentions that, when in the same environment, some herbivore species of fish specifically chose to eat different algae species compared to green sea turtles in order to eliminate competition over food resources. After describing digestion, which is not needed for this study, the author transitions into Anthropogenic effects on foraging ecology and nutrition with a specific focus on marine debris.
Young sea turtles are constantly threatened by floating debris. Percentages of post-hatchling loggerheads ingesting tar and plastic demonstrate how this is a key factor against conservation efforts. More facts are mentioned, making the reader note that small amounts of debris can kill a sea turtle. Despite “mortality resulting from ingestion of debris” being difficult to estimate and assess, the damage that debris causes to sea turtles has been studied and discussed time and time again. The author notes that the “[e]ffect of nutrient dilution on intake is being being evaluated in loggerhead hatchlings” (219). The next section discusses commercial fisheries.
Right of the bat, incidental capture of sea turtles is another damaging factor to their increasing mortality rates. Fishing degrades foraging habitats, specifically trawling can destroy whole communities by decreasing the quantity and quality. Another idea for further study is investigating competition of food resources between humans and sea turtles. Through commercial fishing more and more species of fish are becoming commercially extinct leading to disruptions in food webs (due to population shifts and habitat degradation) as well as decreasing food rates of sea turtles. Furthermore sea grasses grow slowly or not at all within areas covered in scars from anchors. Oil spills also threatened sea turtle species as well as their food distribution.
In closing, the author suggests further research between relationships between foraging ecology and sea turtles. This study needs to involve wild sea turtles and natural diet(s). Overall this chapter was extremely easy to comprehend and analyze for this abstract.
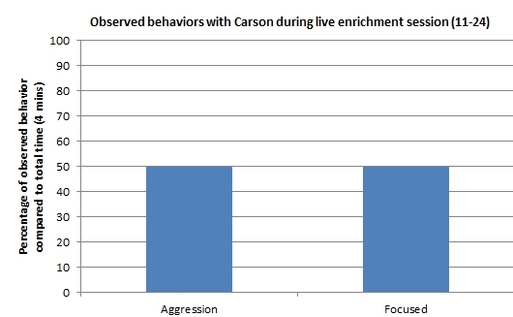
Here is a graphical representation that displays percentages of each behavior observed with Carson, yearling loggerhead, during Saturday's live enrichment session. This displays that for half of the time, Carson was chasing the fish while the other half of the session was spent by aggressively trying to bite it. At the end of the session the shiner perch was eaten.
Note: During this session a tour was surrounding the tank's window, creating a possible distraction for Carson.
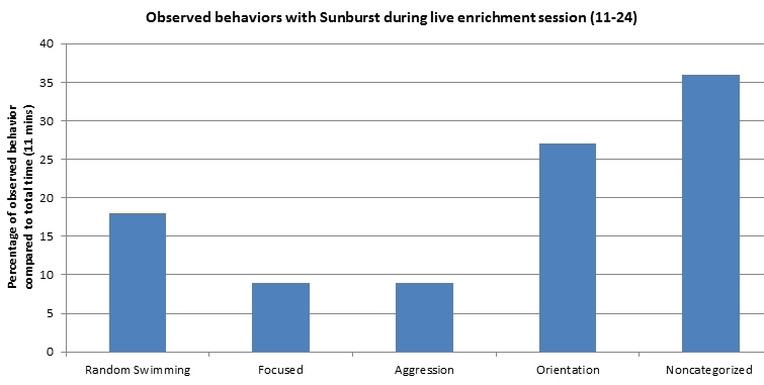
Here is a graphical representation that displays percentages of each behavior observed with Sunburst, sub-adult green, during Saturday's live enrichment session. At the end of the session the shiner perch was eaten.
Note: During this session the sea grass bed and jollyball 'coral' feeders were still present in the tank. Therefore the shiner perch was allowed to swim underneath to hide from Sunburst. Since the other feeding enrichment devices were present, Sunburst spent the majority of the time eating the remaining food from the feeding enrichment devices, making this behavior 'noncategorized' since Sunburst was not interacting with the shiner perch.
Orientation

Here is Carson about to aggressively bite the easy water jug (with six holes) that's filled with surf clam. After the twenty minutes had passed, less than half of the original food amount remained within the enrichment device.
Difficult Salad

Here is the difficult water jug (with four holes) containing Sunburst's food before the session. On Tuesday the sub-adult green ate broccoli and green pepper. After the twenty minutes had passed, the majority of the food was still in the water jug.
Chomp!

After some time Carson's ice treat has melted, allowing it to rip of chunks of smelt from the ice cube.
Orientation

Sunburst, sub-adult green, orienting to its frozen treat of romaine at the beginning of the session.
Frozen Enrichment

Carson, yearling loggerhead, about to aggressively attempt to chomp down on its first large-paint bucket-size ice treat filled with frozen smelt.
Food choice, reinforcer preference, and visual discrimination in Monitor Lizards Abstract
Gaalema, Dianna E. "Food Choice, Reinforcer preference, and visual discrimination in Monitor Lizards (Varanus Spp.).”Thesis. Georgia Institute of Technology, 2007. Georgia Institute of Technology. Web. 19 Nov. 2012. <http://smartech.gatech.edu/bitstream/handle/1853/19799/gaalema_diann_e_200712_mast.pdf.txt?sequence=2>.
This is a reliable source since it’s a graduate’s thesis presented at the Georgia Institute of Technology This purpose of this study was to determine what monitor lizards’ food preferences are (i.e size, smell, etc). The author begins her thesis by discussing the history of reptile studies. Later Gaalema suggests that olfactory, movement, and location should play a role when observing foraging behaviors of an active animal. She then makes a specific connection between turtles and monitor lizards- they are easy to observe foraging behaviors. Through citing a couple of studies, Gaalema also notes that “[m]onitor lizards are highly food motivated (Gaalema & Benboe, 2005) and have physiological metabolic control similar to mammals, making them more active and responsive than most lizards (Porges, Riniolo, McBride, & Campbell, 2003)”. She also suggests that a reinforcer (aka stimuli) should be used during target training. Similar to the loggerhead sea turtles in my study, all of the lizards involved in these studies are carnivorous. However all of the food was the same size whereas the food differs in weight with my study since there are three different age groups,which all eat varying diets and sizes, present. Gaalema notes that the majority of the lizards were kept by themselves within their enclosures. She decided to use all of the same size for the lizards’ food since previous studies have shown that monitor lizards base their food preference on size. Furthermore due to the fact that this study is investigating food preference of monitor lizards, multiple food items were placed within each lizard’s enclosure whereas in my study only one food item is placed within the tank at a time. The food that was used within this study included mice, fish, eggs, chicks, and crickets. All of the lizards had a higher preference to mice compared to the other food items granted to the individual in question. Each session lasted until the individual did not respond for 60 seconds. The team’s hypothesis was correct- the lizards preferred mice than quail eggs. The author suggests further investigation by testing out different variables (i.e food, water, heat, escape, and changes in illumination) against each other in order to assess effectiveness. The author notes that “a highly preferred item could be used to encourage interaction with a particular enrichment device (Tarou & Bashaw, 2007)”. Therefore this technique could be used in the case of Sunburst in terms of leaving in her sea grass bed in favor of chasing and eating live food.
Within the general discussion, the author suggests further study either by testing olfactory discrimination, “demonstrat[ing the] concept of learning in these animals”, as well as discovering difference(s) between monitor lizards and other animals. She then encourages zoos to conduct research on reptiles since they tend to have a diverse collection within their herpetology departments; “Like Card et al., Hosey states that zoos are not reaching their research potential, and that research conducted at zoos, especially basic research, should be increased. As such, increasing the amount of basic research that is done in zoos and the dissemination of the results to the herpetological world as well as the zoo world should be encouraged.”
Only the first experiment presented was discussed within this abstract since the second one doesn’t connect to my study in addition to the last experiment being difficult to read and analyze due to it’s confusing phrasing.
This is a reliable source since it’s a graduate’s thesis presented at the Georgia Institute of Technology This purpose of this study was to determine what monitor lizards’ food preferences are (i.e size, smell, etc). The author begins her thesis by discussing the history of reptile studies. Later Gaalema suggests that olfactory, movement, and location should play a role when observing foraging behaviors of an active animal. She then makes a specific connection between turtles and monitor lizards- they are easy to observe foraging behaviors. Through citing a couple of studies, Gaalema also notes that “[m]onitor lizards are highly food motivated (Gaalema & Benboe, 2005) and have physiological metabolic control similar to mammals, making them more active and responsive than most lizards (Porges, Riniolo, McBride, & Campbell, 2003)”. She also suggests that a reinforcer (aka stimuli) should be used during target training. Similar to the loggerhead sea turtles in my study, all of the lizards involved in these studies are carnivorous. However all of the food was the same size whereas the food differs in weight with my study since there are three different age groups,which all eat varying diets and sizes, present. Gaalema notes that the majority of the lizards were kept by themselves within their enclosures. She decided to use all of the same size for the lizards’ food since previous studies have shown that monitor lizards base their food preference on size. Furthermore due to the fact that this study is investigating food preference of monitor lizards, multiple food items were placed within each lizard’s enclosure whereas in my study only one food item is placed within the tank at a time. The food that was used within this study included mice, fish, eggs, chicks, and crickets. All of the lizards had a higher preference to mice compared to the other food items granted to the individual in question. Each session lasted until the individual did not respond for 60 seconds. The team’s hypothesis was correct- the lizards preferred mice than quail eggs. The author suggests further investigation by testing out different variables (i.e food, water, heat, escape, and changes in illumination) against each other in order to assess effectiveness. The author notes that “a highly preferred item could be used to encourage interaction with a particular enrichment device (Tarou & Bashaw, 2007)”. Therefore this technique could be used in the case of Sunburst in terms of leaving in her sea grass bed in favor of chasing and eating live food.
Within the general discussion, the author suggests further study either by testing olfactory discrimination, “demonstrat[ing the] concept of learning in these animals”, as well as discovering difference(s) between monitor lizards and other animals. She then encourages zoos to conduct research on reptiles since they tend to have a diverse collection within their herpetology departments; “Like Card et al., Hosey states that zoos are not reaching their research potential, and that research conducted at zoos, especially basic research, should be increased. As such, increasing the amount of basic research that is done in zoos and the dissemination of the results to the herpetological world as well as the zoo world should be encouraged.”
Only the first experiment presented was discussed within this abstract since the second one doesn’t connect to my study in addition to the last experiment being difficult to read and analyze due to it’s confusing phrasing.
Aftermath

Here is Ghostbuster, after the video below was taken, with the other half of the crab remaining after the >1 minute live enrichment session.
Saturday's live enrichment session with Ghostbuster (Oct. 27, 2012)
Gulp!

Clemente, yearling loggerhead, displayed going in for another bite. Throughout the whole session Clemente went about taking off each of the crab's legs leaving behind its carapace (which was later given to Ghostbuster).
Saturday's live enrichment session with Clemente (Oct. 27, 2012)
Avoidence

Sunburst, sub-adult green, pictured ignoring its live blue crab (taken after the video below).
Saturday's live enrichment session with Sunburst (Oct. 27, 2012)
Lonely blue crab

One out of the four blue crabs, with claws still intact, before being offered during the live enrichment sessions.
"Different Strokes: Can Managing Behavioral Types Increase Post-release Success?" Abstract
Watters, Jason V., and Cheryl L. Meehan. "Different Strokes: Can Managing Behavioral Types Increase Post-release Success?" Applied Animal Behavior Science 102 (2007): 364-79. 2007. Web. 28 Oct. 2012. <http://homepage.psy.utexas.edu/homepage/group/AnimPersInst/Animal%20Personality
%20PDFs/W/Watters%20%26%20Meehan%202007.pdf>.
This is a reliable source since both of the authors are associated with University of California. The article begins by informing the reader of possible ways in which release programs might fail. The main one being that the animal(s) in question aren’t able to handle foraging techniques in their natural habitat and/or don’t know how to respond effectively with predators. A way to minimize the occurrences of these problems is to make sure that the animal(s) in question are being enriched in a variety of ways rather than just focusing on foraging techniques while ignoring the idea of predators. By not doing this, the animal(s) gain a “high[er] probability of survival in the natural environment (McPhee and Silverman, 2004)” (365). Furthermore this allows staff to assess the animal’s behavior within multiple contexts. The authors then discuss the possibility of behavioral types, such as excelling in avoidance of predators despite also not efficiently foraging. In other words;
“some individuals may be ‘shy’ while others are ‘bold’ (Coleman and Wilson, 1998; Wilson et al., 1994). Bold individuals may be more likely to
sample novel foods (Kelley et al., 2005) or they may maintain a high activity schedule, even in the presence of predators. On the other hand,
shy individuals might maintain low activity levels and feed less but simultaneously have a lower overall predation risk” (365).
Another additional benefit of being ‘shy’ is being able to notice environmental changes; “Apparently these individuals rely more on external cues to determine their responses to the environment while bold individuals act in a manner more intrinsically driven and routine-like (Marchetti and Drent, 2000)” (365). In addition to different personalities, each individual may act differently due to either their phenotype(s) (aka not enough genetic variation) or lacking “a fully developed range of species-typical behaviors” (366) . In other words, in terms of discussing phenotypes; “it may be difficult for individuals who have followed a specific developmental trajectory to express the behaviors that might develop along an alternative trajectory. In such cases, environmental variation at the population level is important in the expression of individual behavioral types” (366). An individual’s physiology may also play a role in being able to survive in their natural habitat, as in different physiological differences might allow an individual to be vulnerable to various pathogens. The authors then relate this concept back to the personality differences in terms of “bold[er individuals] appear to be more susceptible to autoimmune disease than shy individuals (Hessing et al., 1995)” (366). In order to successfully release a group of individuals back into their natural habitat, the authors suggest to assess all of these factors: personality, phenotypes, and whether or not they have fully developed a range of species-typical behaviors. Specifically the reader(s) should “determine and promote the expression of individuals’ behavioral types” (366). In addition to this the reader(s) need to “understand their causes, determine effective means of assaying individuals for their behavioral type, and develop protocols for directing the expression of behavioral types in animals” (366). The authors then discuss social groups, which doesn’t apply to my study since sea turtles are solitary creatures.
The paper transitions into talking about phenotypes and genetic diversity within various populations (within the animal’s natural environment as well as their captive one). Since captive environments have a limited amount of animals as well as participating in a specific breeding program, these zoological institutions tend to have low genetic variety between individuals; “As a result, the few animals that are entered into captive rearing programs are a precious commodity because they represent the small potential to rebuild wild populations” (368). The authors then note how some captive programs purposefully adjust the animal’s environment and experiences in order “to promote the expression of variant behavioral types within the range possible for the genotypes in the captive population” (368). The paper then transitions to discussing how different phenotypes may also play a role within reproductive success in animals, which is key to getting sea turtles removed from the endangered species list. In part of this, when competition is removed, whether reproductive or hunting techniques, the animal(s) in question may develop different behavior compared to being within their natural habitat due to zoological institutions limiting or eliminating competition altogether (which is why my study also focuses on live enrichment where the individual is forced to hunt prey). At the end of this section, the authors suggest to the reader to also consider “delivering a varied, relevant set of developmental circumstances that drive the expression of variant behavioral types (Watters et al., 2003)” (369). The next section discussing assessing behaviors within a captive setting.
The authors note that animals within a captive setting give researchers a unique opportunity to examine behavior(s) as well as understanding how the animal(s) relate to their “housing, husbandry and other environmental factors [which] may impact [the] well-being and reproductive success [of the animal(s)] (Carlstead et al., 1999)” (369). The paper then transitions into discussion of environmental enrichment in the context of behavioral types, which is the focus of my study. They note that “pre-release environmental enrichment [allows animals to develop behavioral skills necessary for survival] through the application of strategies (e.g. Miller et al., 1998)” (370). It is then suggested, by citing various studies, that environmental enrichment is also used as a way to assess behavioral types of captive animals (in my case foraging behaviors, as defined within “Experimental Evaluation of Environmental Enrichment of Sea Turtles” Abstract). The authors then suggest a way to observe these behavioral differences, which I’m already doing within my study; “[inspect the] duration of interaction with devices and the quality of interactions (vigorous, tentative,etc.)” (371). The paper later discussing using environmental enrichment as a way to assess foraging behavior(s), which again relates to my study;
“Such tasks may engage the animal in a type of problem solving and often animals quickly become proficient at completing foraging tasks to
obtain food items. By providing an array of foraging tasks to members of captive populations and observing the response of individual animals,
managers can assess the range of behavioral responses within the group” (372).
The authors also note that “some individuals may vigorously interact with foraging devices and learn to access food items through repeated trial and error [(such as the loggerhead patients)], while others play a more observant role [(such as the sub-adult green sea turtle)]” (372). They also suggest to their readers that they should assess captive behavior (types) more accurately by “diversify[ing] the types of foraging styles that release groups express” (373). The need for predation as well as competition of captive animals are then discussed. In terms of competition, feeding device use is highly recommended since it “require[s] animals to visit specific sites and perform foraging tasks to retrieve food items provide an excellent opportunity for researchers to observe individuals within the group as they adopt strategies to access the resource” (374; which is currently being tested with the sub-adult green through the use of jollyball ‘coral’ feeders).
Within the discussion the authors note a couple of ways in which further study should be conducted, the first being able to “determine the potential of, and best methods for, application of phenotype management that focuses on behavioral types in the context of captive rearing programs” (375). The first step in undergoing with this research, is to assess behavioral types under specific environmental conditions. They also suggest that “[a]dditional work should follow animals of different types after release and determine their survival and reproductive success in relation to multiple ecological contexts [such as varied predation risks, interaction with conspecifics, food availability, distribution and type, habitat characteristics, environmental perturbation, etc]” (375), which cannot be done with the Pittsburgh Zoo & PPG Aquarium’s turtles since satellite transmitters only relay information for approximately eight months, keeping in mind that sea turtles don’t sexually mature until around the age of twenty.
Overall this article was lengthy compared to other articles and blog posts abstracted in the past. In addition to this, the wording was difficult to understand since it was written for those within zoological fields of study. Furthermore, the article primarily focused on populations, phenotypes, and social animals while my study doesn’t have nearly enough time and financial resources to investigate phenotypes of all four of the patients as well as, again, sea turtles being solitary creatures.
%20PDFs/W/Watters%20%26%20Meehan%202007.pdf>.
This is a reliable source since both of the authors are associated with University of California. The article begins by informing the reader of possible ways in which release programs might fail. The main one being that the animal(s) in question aren’t able to handle foraging techniques in their natural habitat and/or don’t know how to respond effectively with predators. A way to minimize the occurrences of these problems is to make sure that the animal(s) in question are being enriched in a variety of ways rather than just focusing on foraging techniques while ignoring the idea of predators. By not doing this, the animal(s) gain a “high[er] probability of survival in the natural environment (McPhee and Silverman, 2004)” (365). Furthermore this allows staff to assess the animal’s behavior within multiple contexts. The authors then discuss the possibility of behavioral types, such as excelling in avoidance of predators despite also not efficiently foraging. In other words;
“some individuals may be ‘shy’ while others are ‘bold’ (Coleman and Wilson, 1998; Wilson et al., 1994). Bold individuals may be more likely to
sample novel foods (Kelley et al., 2005) or they may maintain a high activity schedule, even in the presence of predators. On the other hand,
shy individuals might maintain low activity levels and feed less but simultaneously have a lower overall predation risk” (365).
Another additional benefit of being ‘shy’ is being able to notice environmental changes; “Apparently these individuals rely more on external cues to determine their responses to the environment while bold individuals act in a manner more intrinsically driven and routine-like (Marchetti and Drent, 2000)” (365). In addition to different personalities, each individual may act differently due to either their phenotype(s) (aka not enough genetic variation) or lacking “a fully developed range of species-typical behaviors” (366) . In other words, in terms of discussing phenotypes; “it may be difficult for individuals who have followed a specific developmental trajectory to express the behaviors that might develop along an alternative trajectory. In such cases, environmental variation at the population level is important in the expression of individual behavioral types” (366). An individual’s physiology may also play a role in being able to survive in their natural habitat, as in different physiological differences might allow an individual to be vulnerable to various pathogens. The authors then relate this concept back to the personality differences in terms of “bold[er individuals] appear to be more susceptible to autoimmune disease than shy individuals (Hessing et al., 1995)” (366). In order to successfully release a group of individuals back into their natural habitat, the authors suggest to assess all of these factors: personality, phenotypes, and whether or not they have fully developed a range of species-typical behaviors. Specifically the reader(s) should “determine and promote the expression of individuals’ behavioral types” (366). In addition to this the reader(s) need to “understand their causes, determine effective means of assaying individuals for their behavioral type, and develop protocols for directing the expression of behavioral types in animals” (366). The authors then discuss social groups, which doesn’t apply to my study since sea turtles are solitary creatures.
The paper transitions into talking about phenotypes and genetic diversity within various populations (within the animal’s natural environment as well as their captive one). Since captive environments have a limited amount of animals as well as participating in a specific breeding program, these zoological institutions tend to have low genetic variety between individuals; “As a result, the few animals that are entered into captive rearing programs are a precious commodity because they represent the small potential to rebuild wild populations” (368). The authors then note how some captive programs purposefully adjust the animal’s environment and experiences in order “to promote the expression of variant behavioral types within the range possible for the genotypes in the captive population” (368). The paper then transitions to discussing how different phenotypes may also play a role within reproductive success in animals, which is key to getting sea turtles removed from the endangered species list. In part of this, when competition is removed, whether reproductive or hunting techniques, the animal(s) in question may develop different behavior compared to being within their natural habitat due to zoological institutions limiting or eliminating competition altogether (which is why my study also focuses on live enrichment where the individual is forced to hunt prey). At the end of this section, the authors suggest to the reader to also consider “delivering a varied, relevant set of developmental circumstances that drive the expression of variant behavioral types (Watters et al., 2003)” (369). The next section discussing assessing behaviors within a captive setting.
The authors note that animals within a captive setting give researchers a unique opportunity to examine behavior(s) as well as understanding how the animal(s) relate to their “housing, husbandry and other environmental factors [which] may impact [the] well-being and reproductive success [of the animal(s)] (Carlstead et al., 1999)” (369). The paper then transitions into discussion of environmental enrichment in the context of behavioral types, which is the focus of my study. They note that “pre-release environmental enrichment [allows animals to develop behavioral skills necessary for survival] through the application of strategies (e.g. Miller et al., 1998)” (370). It is then suggested, by citing various studies, that environmental enrichment is also used as a way to assess behavioral types of captive animals (in my case foraging behaviors, as defined within “Experimental Evaluation of Environmental Enrichment of Sea Turtles” Abstract). The authors then suggest a way to observe these behavioral differences, which I’m already doing within my study; “[inspect the] duration of interaction with devices and the quality of interactions (vigorous, tentative,etc.)” (371). The paper later discussing using environmental enrichment as a way to assess foraging behavior(s), which again relates to my study;
“Such tasks may engage the animal in a type of problem solving and often animals quickly become proficient at completing foraging tasks to
obtain food items. By providing an array of foraging tasks to members of captive populations and observing the response of individual animals,
managers can assess the range of behavioral responses within the group” (372).
The authors also note that “some individuals may vigorously interact with foraging devices and learn to access food items through repeated trial and error [(such as the loggerhead patients)], while others play a more observant role [(such as the sub-adult green sea turtle)]” (372). They also suggest to their readers that they should assess captive behavior (types) more accurately by “diversify[ing] the types of foraging styles that release groups express” (373). The need for predation as well as competition of captive animals are then discussed. In terms of competition, feeding device use is highly recommended since it “require[s] animals to visit specific sites and perform foraging tasks to retrieve food items provide an excellent opportunity for researchers to observe individuals within the group as they adopt strategies to access the resource” (374; which is currently being tested with the sub-adult green through the use of jollyball ‘coral’ feeders).
Within the discussion the authors note a couple of ways in which further study should be conducted, the first being able to “determine the potential of, and best methods for, application of phenotype management that focuses on behavioral types in the context of captive rearing programs” (375). The first step in undergoing with this research, is to assess behavioral types under specific environmental conditions. They also suggest that “[a]dditional work should follow animals of different types after release and determine their survival and reproductive success in relation to multiple ecological contexts [such as varied predation risks, interaction with conspecifics, food availability, distribution and type, habitat characteristics, environmental perturbation, etc]” (375), which cannot be done with the Pittsburgh Zoo & PPG Aquarium’s turtles since satellite transmitters only relay information for approximately eight months, keeping in mind that sea turtles don’t sexually mature until around the age of twenty.
Overall this article was lengthy compared to other articles and blog posts abstracted in the past. In addition to this, the wording was difficult to understand since it was written for those within zoological fields of study. Furthermore, the article primarily focused on populations, phenotypes, and social animals while my study doesn’t have nearly enough time and financial resources to investigate phenotypes of all four of the patients as well as, again, sea turtles being solitary creatures.
Here is a graphical representation that displays percentages of each behavior observed with Sunburst, sub-adult green, during Saturday's live enrichment session. At the end of the session all three feeder guppies were still alive.
Here is a graphical representation that displays percentages of each behavior observed with Ghostbuster, juvenile loggerhead, during Saturday's live enrichment session. At the end of the session all three feeder guppies were eaten.
Note: unlike all of the other graphical displays Ghosbuster ate all of the feeder guppies, thus the session ended at approximately seven minutes compared to the usual twenty minute session.
Note: unlike all of the other graphical displays Ghosbuster ate all of the feeder guppies, thus the session ended at approximately seven minutes compared to the usual twenty minute session.
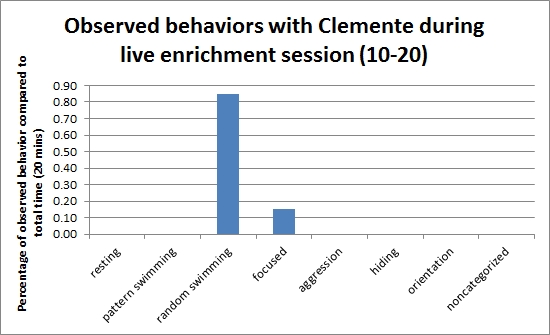
Here is a graphical representation that displays percentages of each behavior observed with Clemente, yearling loggerhead, during Saturday's live enrichment session. At the end of the session all three feeder guppies were still alive.
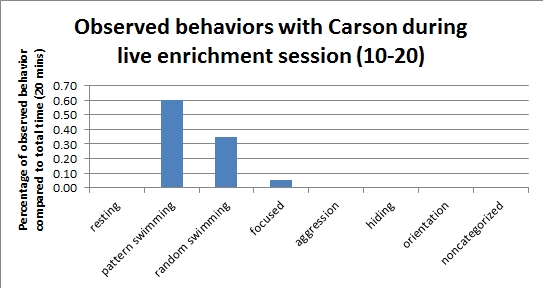
Here is a graphical representation that displays percentages of each behavior observed with Carson, yearling loggerhead, during Saturday's live enrichment session. At the end of the session all four feeder guppies were still alive.
Another type of (unrelated) enrichment

Here is Sunburst's back-scratcher that is constantly used by the turtle in order to rub off dead keratin from its carapace, therefore the chance of lesions of build-up of dead keratin appearing is decreased. Similar to this enrichment device all of the loggerhead patients as well as Sunburst have spray bars located within all of their tanks.
Another frozen treat

Clemente, yearling loggerhead, about to devour its frozen smelt.
Thursday's frozen enrichment session with Clemente (Oct. 18, 2012)
More frozen smelt

Ghostburster eating more of its frozen smelt.
Frozen Enrichment

Ghostbuster eating its frozen smelt after the 'frozen treat' melted.
Thursday's frozen enrichment session with Sunburst (Oct. 18, 2012)
Monterey Bay Aquarium blog post abstract
Melissa. "Sea Turtle Enrichment." Monterey Bay Aquarium, 11 Dec. 2009. Web. 17 Oct. 2012. <http://montereybayaquarist.blogspot.com/search?q=enrichment>.
This blog post is extremely basic and straightforward. The article has a primary focus on back scratchers for sea turtles as well as briefly discussing food-based enrichment. Ice blocks, bull kelp, and pumpkins are given to their rehab patients. For people such as myself looking for substantial evidence that validates my methods this blog post is adequate. However at this point I’m looking for various new ideas to incorporate into my methods and to see what others within the discipline of sea turtle rehabilitation are doing, in that sense this article barely scratches the surface.
This blog post is extremely basic and straightforward. The article has a primary focus on back scratchers for sea turtles as well as briefly discussing food-based enrichment. Ice blocks, bull kelp, and pumpkins are given to their rehab patients. For people such as myself looking for substantial evidence that validates my methods this blog post is adequate. However at this point I’m looking for various new ideas to incorporate into my methods and to see what others within the discipline of sea turtle rehabilitation are doing, in that sense this article barely scratches the surface.
Tuesday's feeding device enrichment session with Ghostbuster (Oct. 16, 2012)
Jollyball enrichment

Here is Carson, a yearling loggerhead, uninterested in its jollyball that is filled with shrimp.
Sea grass bed enrichment

Here is Sunburst's sea grass bed covered in endive, romaine, and some car wash strips. It's amazing how instantly Sunburst relaxes after this is placed within its tank.
Juvenile water jug enrichment

Ghostbuster attempting to receive its shrimp from a water jug with four holes.
Saturday's live enrichment session with Ghostbuster (Oct. 13, 2012)
Yearling live enrichment

One of the yearlings chasing its live feeder guppy.
Juvenile live enrichment

Here is Ghostbuster chasing its live shiner perch.
Jollyball 'coral' feeder in action

Sunburst successfully using new jollyball 'coral' feeder.
Another jollyball 'coral' feeder

Here is another new feeding device that's covered in holes rather than string, allowing carrot and cucumber slices to stick out for Sunburst to eat.
First jollyball 'coral' feeder

Here is a new feeding enrichment device created by Josie and some interns. Half a jollyball laced with string (bottom attached to a weight) that's laced with endive, one of Sunburst's favorite greens.
Yearling Frozen Enrichment

Here is Clemente, a yearling loggerhead, investigating its frozen shrimp treat. It was gone in approximately 3 minutes.
More Frozen Enrichment

Ghostbuster about to eat frozen smelt.
Thursday's frozen enrichment session with Ghostbuster (Oct. 11, 2012)
Frozen Enrichment

Ghostbuster pictured while displaying focused behavior; "Investigation of enrichment device- includes biting, pushing, and rubbing against item" (Therrien et al.’s, 2007).
"Turtle Enrichment with a Twist" Abstract
"Turtle Enrichment with a Twist." Georgia Sea Turtle Center, 2011. Web. 11 Oct. 2012. <http://www.georgiaseaturtlecenter.org/wp-content/uploads/2012/08/Spring-2011.pdf>.
This newsletter article within Georgia Sea Turtle Center’s newsletter published in the spring of 2011 begins with a basic overall definition of enrichment. Similar to my project this center also only focuses on sea turtle rehabilitation. The article then discusses frozen treats (which is one of the food-based enrichment that I’m currently observing) and benefits that sea turtles gain from this enrichment; “Freezing [food]...in blocks of ice is a form of enrichment for sea turtles in active recovery”. The article then notes that frozen treats are both a mental and physical challenge due to the difficulty of grasping with their beak as well as chasing. By giving rehab turtles frozen treats, it’s mimicking their natural diet of hard-shelled animals (such as whelks, blue crabs, and horseshoe crabs), “[t]hus, ice block enrichment has the added benefit of natural beak trimming. Some turtles have even learned to wait for the ice to melt and food to fall to the bottom of the tank[(which has been observed with Ghostbuster)]!” The article then discusses live food enrichment (such as live blue and fiddler crabs) and how the patient must be healthy enough in order to chase and properly consume the said creature. The end of the article ends on a great quote from one of the Rehabilitation Technicians; “The ultimate goal for all animals admitted to the Georgia Sea Turtle Center is a successful released back to the wild. We try to keep them as wild as possible throughout their recovery and enrichment items are invaluable in encouraging natural behaviors.”
Overall this newsletter article was easy to understand. It was good to be reassured that live, frozen, and feeding devices are perfect types of enrichment for rehabilitating turtles (both loggerheads and greens). However I wished that they would have briefly discussed behaviors that are observed during food-based enrichment.
This newsletter article within Georgia Sea Turtle Center’s newsletter published in the spring of 2011 begins with a basic overall definition of enrichment. Similar to my project this center also only focuses on sea turtle rehabilitation. The article then discusses frozen treats (which is one of the food-based enrichment that I’m currently observing) and benefits that sea turtles gain from this enrichment; “Freezing [food]...in blocks of ice is a form of enrichment for sea turtles in active recovery”. The article then notes that frozen treats are both a mental and physical challenge due to the difficulty of grasping with their beak as well as chasing. By giving rehab turtles frozen treats, it’s mimicking their natural diet of hard-shelled animals (such as whelks, blue crabs, and horseshoe crabs), “[t]hus, ice block enrichment has the added benefit of natural beak trimming. Some turtles have even learned to wait for the ice to melt and food to fall to the bottom of the tank[(which has been observed with Ghostbuster)]!” The article then discusses live food enrichment (such as live blue and fiddler crabs) and how the patient must be healthy enough in order to chase and properly consume the said creature. The end of the article ends on a great quote from one of the Rehabilitation Technicians; “The ultimate goal for all animals admitted to the Georgia Sea Turtle Center is a successful released back to the wild. We try to keep them as wild as possible throughout their recovery and enrichment items are invaluable in encouraging natural behaviors.”
Overall this newsletter article was easy to understand. It was good to be reassured that live, frozen, and feeding devices are perfect types of enrichment for rehabilitating turtles (both loggerheads and greens). However I wished that they would have briefly discussed behaviors that are observed during food-based enrichment.
Saturday's live enrichment session with Ghostbuster (Oct. 6, 2012)
"Environmental Enrichment for Sea Turtles in Rehabilitation: Preliminary Study" Abstract
Lloyd, Janice, Ellen Ariel, Dayna Adams, and Leigh Owens. "Environmental Enrichment for Sea Turtles in Rehabilitation: Preliminary Study." Web. 1 Oct. 2012. <http://www.awrc.org.au/uploads/5/8/6/6/5866843/ariel_lloyd.pdf>.
The authors begin their introduction by stating that since 2009 Queensland, AU has had tons of rain. Due to the excessive rain fall, sea grasses were not exposed to the necessary amount of sunlight; as a result, due to starvation, sea turtles became stranded. The authors then compare natural environments with the sterile environments that sea turtles are placed into during the rehabilitation process. They also note there is limited research that discusses enrichment studies conducted on reptiles. By citing another study Lloyd and her team discuss how enrichment is as effective with marine reptiles as it is with other animals, therefore sea turtles should gain similar benefits.
Within the methods section the authors note that the four green sea turtles’ ages and sexes were unknown at the time of this study. Since all of these turtles suffered from starvation, they required special enrichment; “we added a range of enrichment devices, both sunken and floating, to the each of the turtles’ tanks for a period of 20 minutes. These devices included: balls, pipes, waterfall, boxes, brooms and food dispensing devices” (1). They used Therrien et al.’s (2007) behavioral definitions (which is what is being used in my study). The authors predicted that these enrichment devices would decrease the amount of pattern swimming and resting behavior observed.
In the end the authors discovered that individual behavior differed “...significantly ... from each other depending on what enrichment device was present. Trends in the dataset suggest that pattern swimming and resting behaviour were reduced when enrichment devices were present” (2). Within the discussion the authors note how enrichment is an inexpensive way to maintain the wellbeing of sea turtles that are being rehabilitated. They suggest further study using a larger variety of enrichment devices since the four greens’ differed with each device. In addition to this, “Further research [should] be conducted to look at the effects of specific enrichment devices on behaviour; it would be interesting to look at colour and size of objects that were not controlled for in this preliminary study” (2).
Overall this article was easy to comprehend. However the authors do not specify which type of enrichment they are testing in addition to the ages of the case studies being unknown. In addition to this, this study focuses on four green sea turtles while our study is investigating loggerheads (primarily at the post-hatchling age).
The authors begin their introduction by stating that since 2009 Queensland, AU has had tons of rain. Due to the excessive rain fall, sea grasses were not exposed to the necessary amount of sunlight; as a result, due to starvation, sea turtles became stranded. The authors then compare natural environments with the sterile environments that sea turtles are placed into during the rehabilitation process. They also note there is limited research that discusses enrichment studies conducted on reptiles. By citing another study Lloyd and her team discuss how enrichment is as effective with marine reptiles as it is with other animals, therefore sea turtles should gain similar benefits.
Within the methods section the authors note that the four green sea turtles’ ages and sexes were unknown at the time of this study. Since all of these turtles suffered from starvation, they required special enrichment; “we added a range of enrichment devices, both sunken and floating, to the each of the turtles’ tanks for a period of 20 minutes. These devices included: balls, pipes, waterfall, boxes, brooms and food dispensing devices” (1). They used Therrien et al.’s (2007) behavioral definitions (which is what is being used in my study). The authors predicted that these enrichment devices would decrease the amount of pattern swimming and resting behavior observed.
In the end the authors discovered that individual behavior differed “...significantly ... from each other depending on what enrichment device was present. Trends in the dataset suggest that pattern swimming and resting behaviour were reduced when enrichment devices were present” (2). Within the discussion the authors note how enrichment is an inexpensive way to maintain the wellbeing of sea turtles that are being rehabilitated. They suggest further study using a larger variety of enrichment devices since the four greens’ differed with each device. In addition to this, “Further research [should] be conducted to look at the effects of specific enrichment devices on behaviour; it would be interesting to look at colour and size of objects that were not controlled for in this preliminary study” (2).
Overall this article was easy to comprehend. However the authors do not specify which type of enrichment they are testing in addition to the ages of the case studies being unknown. In addition to this, this study focuses on four green sea turtles while our study is investigating loggerheads (primarily at the post-hatchling age).
"The Effect of a Simple Feeding Enrichment Strategy on the Behaviour of Two Asian Small-clawed Otters (Aonyx Cinerea)" Abstract
Ross, Stephen R. "The Effect of a Simple Feeding Enrichment Strategy on the Behaviour of Two Asian Small-clawed Otters (Aonyx Cinerea)." Aquatic Mammals 28.2 (2002): 113-15, 119. 2002. Web.30 Sept. 2012. <http://aquaticmammalsjournal.org/share/AquaticMammalsIssueArchives/2002/AquaticMammals_28-02/28-02_Ross.pdf>.
This is a credible article since Ross is associated with the Lincoln Park zoo in Illinois. He begins his article by discussing carnivore general behavior within a captive setting. He then informs about the three different types of feeding enrichment for carnivores; “...increas[ing] the number of daily feeding sessions (i.e., Shepherdson et al., 1993) to represent a natural behaviour pattern in which animals often spend a high proportion of their activity budget engaged in feeding-related behaviours. The second is to make [an unpredictable] feeding schedule... (i.e., Hawke et al., 2000) to more closely mimic a free ranging situation where the timing of meals is much more variable. The [last] strategy is to make [feeding more difficult]... either by hiding them (Fischbacher & Schmid, 1999) or by adding a level of complexity to the food manipulation process, such as providing live prey (i.e., Shepherdson et al., 1993; Carlstead, 1991) or full carcass meals (Hutchins et al., 1984; Bond & Lindburg, 1990)” (113). Due to food-based enrichment animals exhibit their natural behavior(s). Some benefits include a better welfare “...(Mellen & MacPhee, 2001) and natural history interpretation [becoming] simpler for the zoological institutions (Hutchins et al.,1984)” (113). Similar to loggerheads the free-ranging Asian small-clawed otters (Aonyx cinerea) diet consists of crustaceans, fish, and mollusks. However within a captive setting otters receive pre-processed food resulting in quick consumption and little to no manipulation. The author cites another journal article to prove that giving other mammals this type of food, within a captive setting, the argument can be made that otters may also suffer from negative behavioral effects (113). The author concludes his paragraph by stating his study’s purpose- through food-based enrichment (aka manipulation of food) an effective and inexpensive way for two captive Asian small-clawed otters (Aonyx cinerea) will be determined. He also notes that the “immediate effect of enrichment [and] whether behavioral changes persist[ed] over time and even whether any effect of this new feeding method is generalized to times after the grapevine balls were removed” (114) were also tested.
The author begins his methods and materials section by stating the exhibit’s dimensions. The author then discusses how data was collected, the parameters of the grapevine enrichment device, and the different phases of data collection. Next an ethogram is displayed defining various behaviors. Data was analyzed by “compar[ing] the otters’ behaviour in the baseline phase to that of the three subsequent phases [as well as] calculat[ing] the mean time spent by the male and female in each behavioural category for each phase each week” (115). The author concluded that after introducing the grapevine enrichment device, manipulation of enrichment devices increased significantly. The results of the study display significant behavioral changes for both otters due to food-based enrichment. The author encourages others to create their exhibits enriching for the captive animals. He also suggests further study in order “to provide a more representative sample across different categories of age, sex, rearing history, and captive environment” (119).
The main differences between my loggerhead study and this journal article include: the two otters housed together (since birth), were between ten and eleven years old, the data was collected over the course of a year (“collected 3–6 times daily during 15-min sessions” (114)), as well as data collected from a group of thirteen trained observers. Despite these differences, I found the three approaches to food-based enrichment (for carnivores), at the beginning of the article, enlightening. In terms of the figures displayed throughout the article, the ethograms and graphs were easy to comprehend while the table of data was slightly confusing. After reading this article, it is more evident that food-based enrichment is needed for marine animals (as well as animals in general) in order to stimulate them within a captive environment in addition to improving their overall well-being.
This is a credible article since Ross is associated with the Lincoln Park zoo in Illinois. He begins his article by discussing carnivore general behavior within a captive setting. He then informs about the three different types of feeding enrichment for carnivores; “...increas[ing] the number of daily feeding sessions (i.e., Shepherdson et al., 1993) to represent a natural behaviour pattern in which animals often spend a high proportion of their activity budget engaged in feeding-related behaviours. The second is to make [an unpredictable] feeding schedule... (i.e., Hawke et al., 2000) to more closely mimic a free ranging situation where the timing of meals is much more variable. The [last] strategy is to make [feeding more difficult]... either by hiding them (Fischbacher & Schmid, 1999) or by adding a level of complexity to the food manipulation process, such as providing live prey (i.e., Shepherdson et al., 1993; Carlstead, 1991) or full carcass meals (Hutchins et al., 1984; Bond & Lindburg, 1990)” (113). Due to food-based enrichment animals exhibit their natural behavior(s). Some benefits include a better welfare “...(Mellen & MacPhee, 2001) and natural history interpretation [becoming] simpler for the zoological institutions (Hutchins et al.,1984)” (113). Similar to loggerheads the free-ranging Asian small-clawed otters (Aonyx cinerea) diet consists of crustaceans, fish, and mollusks. However within a captive setting otters receive pre-processed food resulting in quick consumption and little to no manipulation. The author cites another journal article to prove that giving other mammals this type of food, within a captive setting, the argument can be made that otters may also suffer from negative behavioral effects (113). The author concludes his paragraph by stating his study’s purpose- through food-based enrichment (aka manipulation of food) an effective and inexpensive way for two captive Asian small-clawed otters (Aonyx cinerea) will be determined. He also notes that the “immediate effect of enrichment [and] whether behavioral changes persist[ed] over time and even whether any effect of this new feeding method is generalized to times after the grapevine balls were removed” (114) were also tested.
The author begins his methods and materials section by stating the exhibit’s dimensions. The author then discusses how data was collected, the parameters of the grapevine enrichment device, and the different phases of data collection. Next an ethogram is displayed defining various behaviors. Data was analyzed by “compar[ing] the otters’ behaviour in the baseline phase to that of the three subsequent phases [as well as] calculat[ing] the mean time spent by the male and female in each behavioural category for each phase each week” (115). The author concluded that after introducing the grapevine enrichment device, manipulation of enrichment devices increased significantly. The results of the study display significant behavioral changes for both otters due to food-based enrichment. The author encourages others to create their exhibits enriching for the captive animals. He also suggests further study in order “to provide a more representative sample across different categories of age, sex, rearing history, and captive environment” (119).
The main differences between my loggerhead study and this journal article include: the two otters housed together (since birth), were between ten and eleven years old, the data was collected over the course of a year (“collected 3–6 times daily during 15-min sessions” (114)), as well as data collected from a group of thirteen trained observers. Despite these differences, I found the three approaches to food-based enrichment (for carnivores), at the beginning of the article, enlightening. In terms of the figures displayed throughout the article, the ethograms and graphs were easy to comprehend while the table of data was slightly confusing. After reading this article, it is more evident that food-based enrichment is needed for marine animals (as well as animals in general) in order to stimulate them within a captive environment in addition to improving their overall well-being.
Ice treat Enrichment

Romaine frozen for Sunburst, sub-adult green sea turtle.
More Jolly-ball enrichment

Yearling loggerhead interacting with jolly-ball (also filled with shrimp and surf clam chunks).
Jolly-ball Enrichment

Yearling loggerhead receiving its food (shrimp with surf clam junks) in a food-based enrichment device, a jolly-ball.
'Sea grass' bed

Yearling loggerhead interacting with 'sea grass' (car wash stripes attached to PVC pipes) bed enrichment.
Interaction with water Jug Enrichment

Ghostbuster, juvenile loggerhead, receiving his food (shrimp and surf clam chunks) within a water jug.
Water-jug Enrichment
Ghostbuster's, juvenile loggerhead, food (shrimp and surf clam chunks) placed within a water jug.
Scrubbing time!

Here is the resident sub-adult green sea turtle, Sunburst, getting its carapace (shell) scrubbed with a disinfectant solution. This solution helps with the carapace to heal from the boat injury that Sunburst encountered years ago as well as a way to clean it from bacteria and algae.
Green sea turtle's weight

All of the rehab patients also get weighed on a weekly basis.
Green sea turtle's depth measurement

Juvenile length measurement
Juvenile width measurement

Juvenile carapace (shell) length measurement
Yearling carapace (shell) length measurement
The interns also measured the curves of all of the patients' carapaces by using a soft tape measure.
Yearling depth measurement

These measurements were collected by using a pair of calipers.
Yearling width measurement

Feeding Time!

This photo demonstrates how the diets of the sea turtle rehab patients, at the Pittsburgh Zoo and PPG Aquarium, vary based on patient size and diet. The larger container is filled with vegetables for the resident green sea turtle, Sunburst, while the green containers are for the two loggerhead yearlings (Carson and Clemente) and the black container is for the loggerhead juvenile, Ghostbuster. The green and black containers are filled with squid, shrimp, and capelin.
Target Training

Here is Sunburst (green sea turtle) doing some target training in order to ready it to be later placed within the Ocean Exhibit. At this point Sunburst is in an early stage of target training, meaning that its only expected to swim in the general direction of the target in order to get rewarded with food.
Loggerhead about to use PVC pipe enrichment device

Here is Ghostbuster getting a little closer to the PVC pipe.
Loggerhead investigating PVC pipe enrichment device

Here is a loggerhead, Ghostbuster a juvenile loggerhead, with a piece of PVC pipe (primarily used for hiding).
"Companion Animals Symposium: Environmental Enrichment for Companion, Exotic, and Laboratory Animals." Abstract
Morris, C. L., T. Grandin, and N. A. Irlbeck. "Companion Animals Symposium: Environmental Enrichment for Companion, Exotic, and Laboratory Animals." Journal of Animal Science 89.12 (2011): 4227-238. Web. 9 Sept. 2012. <http://grandin.com/references/companion.animal.symposium.2011.pdf>.
This article is a reliable source since Morris is associated with the Omaha’s Henry Doorly Zoo in Omaha, NE. While Dr. Grandin and Irlbeck are associated with the Department of Animal Sciences at Colorado State University in Fort Collins. The authors suggest that by using neuroscience research and literature understanding about animal behavior can improve leading to better environmental enrichment for better animal welfare. Due to limited animal science literature that discusses “the underlying neural mechanisms that drive species specific innate behaviors” (4227). Whereas “neuroscience literature demonstrates that animal brains have emotional systems that are more specific than simply stating an animal exhibits psychological stress” (4227). The authors then define the basic goals of enrichment; “...increas[ing] positive emotions and decreas[ing] time that animals are subjected to fear, panic, and rage” (4228). In addition to this, good enrichment should encourage animals to exhibit “positive emotional systems” (4228). The authors then discuss fear.
An example an animal exhibiting fear is known as fear conditioning; “[where] an animal [is] avoiding a place where it was subjected to a frightening stimulus” (4228). In order to minimize fear and stress, animal caretakers “can determine what emotional system is driving an abnormal behavior, [can lead] to design more effective environmental enrichments [since] the right enrichment would reduce the motivation to keep performing an abnormal behavior” (4229). By citing another source the authors again define the ultimate goals of environmental enrichment; “ Newberry (1995) stated that environmental enrichments should improve the biological function of the animal, and that appropriate endpoints should be measured after application of enrichments. The provided enrichment should prevent abnormal behavior or allow an animal to engage in more normal behaviors (Newberry, 1995)” (4229). In zoos environmental enrichment is also used to “[enhance] animal environments” (4230). However, “[zoological] institutions accredited by the Association of Zoos and Aquariums are required to have a formal written enrichment program that promotes appropriate behavioral opportunities for all species under their care (AZA, 2011). These... standards recommend that an enrichment program be based on current knowledge of species biology, and include a process of planning and approval to ensure animal safety, record-keeping, and assessment” (4231). The AZA also requires these institutions to have designated staff to mandate the enrichment program(s) by managing, applying, and training enrichment (4231). By citing another source, the authors demonstrate that in 2001 there were a lot of improvements that were needed for environmental enrichment programs at zoological institutions was created;
“1) the development of proactive enrichment programs, rather than reactive methods; 2) plans to promote behaviors and activity patterns typical of the species; 3) objective assessment of methods and programs through the application of traditional behavior data collection methods; and 4) promoting accountability for enrichment programs throughout all levels of animal management staff (Mellen and MacPhee, 2001)” (4231).
The authors mention that enrichment helps to educate the public and enhance their experience at zoological institutions. They also suggest more research be completed in order to continue to make more effective enrichment for captive animals.
Within their conclusion the authors discuss the benefits of environmental enrichment programs being practiced successfully compared to the disadvantaged when the staff is using it wrong; “[w]hen environmental enrichment practices are applied correctly, animal husbandry and welfare increase; however, if poorly applied, enrichment practices also can be detrimental to animal welfare” (4235). The authors also stress the need for gaining a better understanding about “the core emotional systems in the brain and their influence on animal behavior [which] will enhance animal husbandry practices” (4235).
This article was incredibly easy to understand. However the authors discussed captive animals as a whole or in terms of primates rather than reptiles, which is what pertains to my enrichment project.
This article is a reliable source since Morris is associated with the Omaha’s Henry Doorly Zoo in Omaha, NE. While Dr. Grandin and Irlbeck are associated with the Department of Animal Sciences at Colorado State University in Fort Collins. The authors suggest that by using neuroscience research and literature understanding about animal behavior can improve leading to better environmental enrichment for better animal welfare. Due to limited animal science literature that discusses “the underlying neural mechanisms that drive species specific innate behaviors” (4227). Whereas “neuroscience literature demonstrates that animal brains have emotional systems that are more specific than simply stating an animal exhibits psychological stress” (4227). The authors then define the basic goals of enrichment; “...increas[ing] positive emotions and decreas[ing] time that animals are subjected to fear, panic, and rage” (4228). In addition to this, good enrichment should encourage animals to exhibit “positive emotional systems” (4228). The authors then discuss fear.
An example an animal exhibiting fear is known as fear conditioning; “[where] an animal [is] avoiding a place where it was subjected to a frightening stimulus” (4228). In order to minimize fear and stress, animal caretakers “can determine what emotional system is driving an abnormal behavior, [can lead] to design more effective environmental enrichments [since] the right enrichment would reduce the motivation to keep performing an abnormal behavior” (4229). By citing another source the authors again define the ultimate goals of environmental enrichment; “ Newberry (1995) stated that environmental enrichments should improve the biological function of the animal, and that appropriate endpoints should be measured after application of enrichments. The provided enrichment should prevent abnormal behavior or allow an animal to engage in more normal behaviors (Newberry, 1995)” (4229). In zoos environmental enrichment is also used to “[enhance] animal environments” (4230). However, “[zoological] institutions accredited by the Association of Zoos and Aquariums are required to have a formal written enrichment program that promotes appropriate behavioral opportunities for all species under their care (AZA, 2011). These... standards recommend that an enrichment program be based on current knowledge of species biology, and include a process of planning and approval to ensure animal safety, record-keeping, and assessment” (4231). The AZA also requires these institutions to have designated staff to mandate the enrichment program(s) by managing, applying, and training enrichment (4231). By citing another source, the authors demonstrate that in 2001 there were a lot of improvements that were needed for environmental enrichment programs at zoological institutions was created;
“1) the development of proactive enrichment programs, rather than reactive methods; 2) plans to promote behaviors and activity patterns typical of the species; 3) objective assessment of methods and programs through the application of traditional behavior data collection methods; and 4) promoting accountability for enrichment programs throughout all levels of animal management staff (Mellen and MacPhee, 2001)” (4231).
The authors mention that enrichment helps to educate the public and enhance their experience at zoological institutions. They also suggest more research be completed in order to continue to make more effective enrichment for captive animals.
Within their conclusion the authors discuss the benefits of environmental enrichment programs being practiced successfully compared to the disadvantaged when the staff is using it wrong; “[w]hen environmental enrichment practices are applied correctly, animal husbandry and welfare increase; however, if poorly applied, enrichment practices also can be detrimental to animal welfare” (4235). The authors also stress the need for gaining a better understanding about “the core emotional systems in the brain and their influence on animal behavior [which] will enhance animal husbandry practices” (4235).
This article was incredibly easy to understand. However the authors discussed captive animals as a whole or in terms of primates rather than reptiles, which is what pertains to my enrichment project.
"Diet of Oceanic Loggerhead Sea Turtles (Caretta Caretta) in the Central North Pacific." Abstract
Parker, Denise M., William J. Cooke, and George H. Balazs. "Diet of Oceanic Loggerhead Sea Turtles (Caretta Caretta) in the Central North Pacific." Fishery Bulletin(2005): 142-52. EBSCOhost. Web. 3 Sept. 2012. <http://content.ebscohost.com/pdf25_26/pdf/hww/03228/p5aqq/ef4.pdfT=P&P=AN&K=506533082&S=R&D=scf&EbscoContent=dGJyMMvl7ESeprE40dvuOLCmr0qep7ZSrqu4SLOWxWXS&ContentCustomer=dGJyMPGosEy1rK5KuePfgeyx44Dt6fIA>.
At the time of publication Parker, worked for NOAA while Balazs worked for the Pacific Islands Fisheries Science Center which is associated with the Honolulu Laboratory, therefor this is a credible source. The authors begin by noting where loggerhead (Caretta caretta) sea turtles are most commonly found- tropical waters as well as where they usually migrate to nest. By citing others, the authors suggest that loggerheads migrate within the Atlantic Ocean due to their diet; “One hypothesis discussed in Polovina et al. (2000; 2004) suggests that this species obtains prey items from the subtropical fronts along which they travel. …[A]long the main frontal area where these turtles are commonly encountered...concentrated phytoplankton... collects and attracts a variety oceanic organisms-many of which may be potential prey times, as well as predators, of oceanic-stage loggerhead sea turtles in the Pacific. Polovina et al. (2000, 2004) have suggested that the turtles are foraging along the TZCF [(the transition zone chlorophyll front)]” (142-3). The authors continue their article by mentioning that the amount of time that juvenile loggerheads spend in the oceanic Pacific is unknown while they are known to spend approximately ten years in the oceanic Atlantic, therefore the authors suggest more research to be completed about the foraging ecology of oceanic-stage loggerheads within the Pacific; “Such information can help identify important food resources and foraging areas necessary for guiding decisions regarding the management of endangered sea turtle populations (Bjorndal, 1999)” (143). The authors then make it evident that they performed this study “to determine the diet composition of loggerhead sea turtles from the central North Pacific Ocean and to discuss the possibility of interactions between these turtles and commercial fisheries that may occur as a result of the foraging behavior of these sea turtles” (143). In order to collect data the National Marine Fisheries Service (NMFS) observers ,between 1990 and 1992, obtained 52 dead (from bycatch) loggerhead sea turtles (143). The NMFS observers also “recorded the capture position and sea surface temperature aboard commercial driftnet vessels” (143). After the specimens were obtained the stomachs were removed and frozen for laboratory analysis; “Gross observations of stomach contents were made and the contents were sorted to the lowest identifiable taxonomic level by using a dissecting microscope. Major fauna were identified, quantified by volume, and the percent contribution (to stomach contents) of each major organism was calculated (Forbes, 1999). Presence of jellyfish or other jellies were identified by presence of tentacles, nematocysts, and whole or partial individuals” (143). Through analysis of the stomachs’ contents some common items were “fish eggs (25% of stomachs), salps, amphipods (46% of stomachs), small fish, and plastic items (35% of stomachs, Table 1). Some plastic items included small plastic beads, thin plastic sheets, polypropylene line, and even a small plastic fish, which had been an individual soy sauce container” (144). Due to the long list of plastic contents it is clear that pollution is a major problem that occurs across the central North Pacific Ocean (where the 52 turtles were widely distributed). A couple of the authors’ conclusions were“[t]he size of the turtle did not appear to be a factor in the type of prey ingested” (145) and “[the] unpredictability of prey availability likely contributes to the opportunistic feeding behavior of the loggerhead sea turtle” (146). The authors also stated that “Eighty-three percent of prey items that were recorded were found floating on the surface or were found on floating objects and would also likely be concentrated at convergent fronts such as the TZCF, driven there by the currents and winds (Polovina, et al., 2000; Polovina et al., 2004). It is suggested that this concentration of prey, along the convergent fronts, may be aggregating the loggerhead sea turtles traveling along this area, which are likely foraging on the increased densities of prey (Polovina et al., 2003a)” (146). As well as stating “[a]nother indication that loggerhead sea turtles are opportunistic feeders is the presence of oceanic, mesopelagic fish as prey items...[but b]ecause of the low numbers, it is likely that loggerhead sea turtles ingest only dead or debilitated fish rather than actively hunt and chase such species. The presence of these species also indicates that the turtles may be feeding at night when they would be more likely to encounter the fish during their diel movement” (146). Another conclusion from this article includes “Loggerhead sea turtles may feed by swallowing floating prey whole and also by biting whole prey (or portions off a whole prey) found on large floating objects” (147). The authors also discovered/verified that loggerheads eat crabs as well as loggerheads can forage within deeper waters “if high densities of prey items are present” (147).
Within their conclusion by citing another source, the authors suggest that if fisheries learn “more about the life history of loggerhead sea turtles and understanding more about the movements, foraging behavior, and prey of these turtles are important for making well-informed management decisions because foraging behavior may change as seasons change and as these turtles move through different habitats (Bjorndal, 1997)” (148); this is an issue against conservation efforts since a ton of sea turtle mortalities occur within gillnet, driftnet, trawl, and longline fishing. At the end of the article the authors suggest that [m]ore studies need to be done on the ecology of these turtles so that fishery interactions at all life stages can be addressed and so that a total picture of the life history of this species can be obtained” (149). Table 1 was the most useful in terms of understanding the average diet of a loggerhead. However some places had difficult phrasing. Despite this difficulty I’ve learned more about loggerhead foraging behavior and their diet, which will ultimately help me in the future when I need to focus on figuring the diets for the different age groups (juvenile and yearling).
At the time of publication Parker, worked for NOAA while Balazs worked for the Pacific Islands Fisheries Science Center which is associated with the Honolulu Laboratory, therefor this is a credible source. The authors begin by noting where loggerhead (Caretta caretta) sea turtles are most commonly found- tropical waters as well as where they usually migrate to nest. By citing others, the authors suggest that loggerheads migrate within the Atlantic Ocean due to their diet; “One hypothesis discussed in Polovina et al. (2000; 2004) suggests that this species obtains prey items from the subtropical fronts along which they travel. …[A]long the main frontal area where these turtles are commonly encountered...concentrated phytoplankton... collects and attracts a variety oceanic organisms-many of which may be potential prey times, as well as predators, of oceanic-stage loggerhead sea turtles in the Pacific. Polovina et al. (2000, 2004) have suggested that the turtles are foraging along the TZCF [(the transition zone chlorophyll front)]” (142-3). The authors continue their article by mentioning that the amount of time that juvenile loggerheads spend in the oceanic Pacific is unknown while they are known to spend approximately ten years in the oceanic Atlantic, therefore the authors suggest more research to be completed about the foraging ecology of oceanic-stage loggerheads within the Pacific; “Such information can help identify important food resources and foraging areas necessary for guiding decisions regarding the management of endangered sea turtle populations (Bjorndal, 1999)” (143). The authors then make it evident that they performed this study “to determine the diet composition of loggerhead sea turtles from the central North Pacific Ocean and to discuss the possibility of interactions between these turtles and commercial fisheries that may occur as a result of the foraging behavior of these sea turtles” (143). In order to collect data the National Marine Fisheries Service (NMFS) observers ,between 1990 and 1992, obtained 52 dead (from bycatch) loggerhead sea turtles (143). The NMFS observers also “recorded the capture position and sea surface temperature aboard commercial driftnet vessels” (143). After the specimens were obtained the stomachs were removed and frozen for laboratory analysis; “Gross observations of stomach contents were made and the contents were sorted to the lowest identifiable taxonomic level by using a dissecting microscope. Major fauna were identified, quantified by volume, and the percent contribution (to stomach contents) of each major organism was calculated (Forbes, 1999). Presence of jellyfish or other jellies were identified by presence of tentacles, nematocysts, and whole or partial individuals” (143). Through analysis of the stomachs’ contents some common items were “fish eggs (25% of stomachs), salps, amphipods (46% of stomachs), small fish, and plastic items (35% of stomachs, Table 1). Some plastic items included small plastic beads, thin plastic sheets, polypropylene line, and even a small plastic fish, which had been an individual soy sauce container” (144). Due to the long list of plastic contents it is clear that pollution is a major problem that occurs across the central North Pacific Ocean (where the 52 turtles were widely distributed). A couple of the authors’ conclusions were“[t]he size of the turtle did not appear to be a factor in the type of prey ingested” (145) and “[the] unpredictability of prey availability likely contributes to the opportunistic feeding behavior of the loggerhead sea turtle” (146). The authors also stated that “Eighty-three percent of prey items that were recorded were found floating on the surface or were found on floating objects and would also likely be concentrated at convergent fronts such as the TZCF, driven there by the currents and winds (Polovina, et al., 2000; Polovina et al., 2004). It is suggested that this concentration of prey, along the convergent fronts, may be aggregating the loggerhead sea turtles traveling along this area, which are likely foraging on the increased densities of prey (Polovina et al., 2003a)” (146). As well as stating “[a]nother indication that loggerhead sea turtles are opportunistic feeders is the presence of oceanic, mesopelagic fish as prey items...[but b]ecause of the low numbers, it is likely that loggerhead sea turtles ingest only dead or debilitated fish rather than actively hunt and chase such species. The presence of these species also indicates that the turtles may be feeding at night when they would be more likely to encounter the fish during their diel movement” (146). Another conclusion from this article includes “Loggerhead sea turtles may feed by swallowing floating prey whole and also by biting whole prey (or portions off a whole prey) found on large floating objects” (147). The authors also discovered/verified that loggerheads eat crabs as well as loggerheads can forage within deeper waters “if high densities of prey items are present” (147).
Within their conclusion by citing another source, the authors suggest that if fisheries learn “more about the life history of loggerhead sea turtles and understanding more about the movements, foraging behavior, and prey of these turtles are important for making well-informed management decisions because foraging behavior may change as seasons change and as these turtles move through different habitats (Bjorndal, 1997)” (148); this is an issue against conservation efforts since a ton of sea turtle mortalities occur within gillnet, driftnet, trawl, and longline fishing. At the end of the article the authors suggest that [m]ore studies need to be done on the ecology of these turtles so that fishery interactions at all life stages can be addressed and so that a total picture of the life history of this species can be obtained” (149). Table 1 was the most useful in terms of understanding the average diet of a loggerhead. However some places had difficult phrasing. Despite this difficulty I’ve learned more about loggerhead foraging behavior and their diet, which will ultimately help me in the future when I need to focus on figuring the diets for the different age groups (juvenile and yearling).
"A Veterinary Assessment of the Risks and Benefits of Environmental Enrichment” Abstract
Duncan, Ann E. "A Veterinary Assessment of the Risks and Benefits of Environmental Enrichment” The Shape of Enrichment, 1997. Web. 17 July 2012. <http://www.enrichment.org/MiniWebs/About_EE/duncan_1997.pdf>.
At the time this was written Dr. Duncan was a veterinarian for the Detroit zoo. Within the introduction Dr. Duncan introduces the term environmental enrichment and why zoological institutions use it from entertaining the public to increase physical activity, decrease stress, as well as promote a healthy mental and emotional state of mind for the animals. Dr. Duncan then explains the process of giving a zoo animal enrichment in terms of the animal keepers discussing it with the veterinary staff as well as the curators as a way to make sure that there is little to no harm done to the animal(s) in addition to benefit the animal(s) in order “to improve [their] overall well-being.” (1) This paper is written to investigate the risks and benefits that are associated with environmental enrichment through various sources as well as discussing how to manage the risks that are involved with environmental enrichment.
However, there aren't many published sources that discuss the risks of enrichment. Dr. Duncan discusses how before using enrichment for application several factors have to be assessed such as:
“the natural history of the species, exhibit and holding area design, makeup of animal groupings mixed or single species, breeding status, age, gender), individual animal personalities, anatomical characteristics, size of enrichment item relative to animal, underlying health conditions, body condition, degree of control over animals (ability to gate etc.), past history with enrichment items, natural and captive diet, keeper schedules, budget, acceptance of unnatural enrichment items on exhibit, and discretion of animal managers” (1).
The more zoological institutions share and collaborate about these factors, the more that the risks and benefits of enrichment can be defined. Dr. Duncan then discusses how data is collected in order to measure the behavioral and physiological effects of enrichment. But first the animal(s), in question, behavior has to be observed, without enrichment, in order to understand what natural behavior(s) the animal(s) exhibit while in solitude. On a physiological standpoint several measurements must take place from white blood cell count to hormone indicators of stress (2). Dr. Duncan then discusses the five main conclusions, in terms of how to solve some of the common problems that occur with captive animals and how to improve their well-beings, from research conducted at zoological institutions and laboratories alike (2).
The first conclusion is that some animals might exhibit ‘self-directed behavior’; “Hair plucking and self-biting in primates, feather plucking in birds and tail sucking/chewing in cats are all examples of self-aggressive behaviors” (3). It was shown that by providing a variety of enrichment devices to the animal(s) these behaviors decreased drastically. The second conclusion is that aggression directed towards others is decreased with the use of several different strategies for different animals; in other words “[o]ne study demonstrated a similar degree of positive behavioral change when comparing two feeding strategies; offering food in foraging racks suspended on walls, and scattering food in shavings on the floor.” (3) The third conclusion is that movement increases by either hiding food or placing it within puzzle feeders. The fourth conclusion found that, based on urinary cortisols, small stressed cats respond to their stressful environment with less movement. The referenced studies also concluded that stressed animals tend to decrease reproductive function; “[t]his suggests that enrichment techniques involving
interaction and operant conditioning might provide hidden breeding benefits in some species.” (4) The fifth conclusion discovered that by providing straw and forage to a group of chimpanzees they have “...reduced levels of other abnormal and antagonistic behaviors and higher levels of locomotion and play (Baker, 1997).” (4) Dental health can also improve by providing food enrichment several times a day, depending on the animals. Overall Dr. Duncan states, “[e]nvironmental enrichment is an effective tool for solving specific problems found in captive animals and can be used to increase overall well-being where problems do not exist.” (4)
Next Dr. Duncan demonstrates, through several literary references, why enrichment must be assessed- based on the physiological and behavioral benefits discussed throughout her paper, it is evident that enrichment needs to be part of “husbandry routines instead of as short-term enhancement.” (4) Without assessment of enrichment techniques zookeeper, veterinary staff, and others time will be wasted due to researching and creating enrichment techniques and devices “that are of little interest to animals and have no positive impact on behavior” (4) as well as being ineffective and “do not provide the greatest potential benefit.” (4) The main risk with enrichment devices, that are shared between multiple animals, is that they are not properly cleansed as well as the risk of animals ingesting the device(s). Dr. Duncan provides a table of eight ways to prevent injury, and even death, while animals are using enrichment techniques and devices. After the table Dr. Duncan discusses that in order to maximize the benefits and minimise the risks of an enrichment technique or device then a zookeeper and the veterinary staff should create effective enrichment for the animals by discussing enrichment applications and possible health risks that might occur. In conclusion Dr. Duncan again states that environmental enrichment provides physiological and behavioral well-being and must continue to be demonstrated to the public as well as animal welfare organizations in order to increase understanding and appreciation of zoo animals being that they are ambassadors to wild creatures.
Overall I thought this paper was easy to understand despite the fourth and fifth conclusions which used quite a lot of veterinary medical terminology. The table that was included was incredibly easy to understand. Based on primarily on the table I now have a better understanding of the potential risks that are involved with environmental enrichment
At the time this was written Dr. Duncan was a veterinarian for the Detroit zoo. Within the introduction Dr. Duncan introduces the term environmental enrichment and why zoological institutions use it from entertaining the public to increase physical activity, decrease stress, as well as promote a healthy mental and emotional state of mind for the animals. Dr. Duncan then explains the process of giving a zoo animal enrichment in terms of the animal keepers discussing it with the veterinary staff as well as the curators as a way to make sure that there is little to no harm done to the animal(s) in addition to benefit the animal(s) in order “to improve [their] overall well-being.” (1) This paper is written to investigate the risks and benefits that are associated with environmental enrichment through various sources as well as discussing how to manage the risks that are involved with environmental enrichment.
However, there aren't many published sources that discuss the risks of enrichment. Dr. Duncan discusses how before using enrichment for application several factors have to be assessed such as:
“the natural history of the species, exhibit and holding area design, makeup of animal groupings mixed or single species, breeding status, age, gender), individual animal personalities, anatomical characteristics, size of enrichment item relative to animal, underlying health conditions, body condition, degree of control over animals (ability to gate etc.), past history with enrichment items, natural and captive diet, keeper schedules, budget, acceptance of unnatural enrichment items on exhibit, and discretion of animal managers” (1).
The more zoological institutions share and collaborate about these factors, the more that the risks and benefits of enrichment can be defined. Dr. Duncan then discusses how data is collected in order to measure the behavioral and physiological effects of enrichment. But first the animal(s), in question, behavior has to be observed, without enrichment, in order to understand what natural behavior(s) the animal(s) exhibit while in solitude. On a physiological standpoint several measurements must take place from white blood cell count to hormone indicators of stress (2). Dr. Duncan then discusses the five main conclusions, in terms of how to solve some of the common problems that occur with captive animals and how to improve their well-beings, from research conducted at zoological institutions and laboratories alike (2).
The first conclusion is that some animals might exhibit ‘self-directed behavior’; “Hair plucking and self-biting in primates, feather plucking in birds and tail sucking/chewing in cats are all examples of self-aggressive behaviors” (3). It was shown that by providing a variety of enrichment devices to the animal(s) these behaviors decreased drastically. The second conclusion is that aggression directed towards others is decreased with the use of several different strategies for different animals; in other words “[o]ne study demonstrated a similar degree of positive behavioral change when comparing two feeding strategies; offering food in foraging racks suspended on walls, and scattering food in shavings on the floor.” (3) The third conclusion is that movement increases by either hiding food or placing it within puzzle feeders. The fourth conclusion found that, based on urinary cortisols, small stressed cats respond to their stressful environment with less movement. The referenced studies also concluded that stressed animals tend to decrease reproductive function; “[t]his suggests that enrichment techniques involving
interaction and operant conditioning might provide hidden breeding benefits in some species.” (4) The fifth conclusion discovered that by providing straw and forage to a group of chimpanzees they have “...reduced levels of other abnormal and antagonistic behaviors and higher levels of locomotion and play (Baker, 1997).” (4) Dental health can also improve by providing food enrichment several times a day, depending on the animals. Overall Dr. Duncan states, “[e]nvironmental enrichment is an effective tool for solving specific problems found in captive animals and can be used to increase overall well-being where problems do not exist.” (4)
Next Dr. Duncan demonstrates, through several literary references, why enrichment must be assessed- based on the physiological and behavioral benefits discussed throughout her paper, it is evident that enrichment needs to be part of “husbandry routines instead of as short-term enhancement.” (4) Without assessment of enrichment techniques zookeeper, veterinary staff, and others time will be wasted due to researching and creating enrichment techniques and devices “that are of little interest to animals and have no positive impact on behavior” (4) as well as being ineffective and “do not provide the greatest potential benefit.” (4) The main risk with enrichment devices, that are shared between multiple animals, is that they are not properly cleansed as well as the risk of animals ingesting the device(s). Dr. Duncan provides a table of eight ways to prevent injury, and even death, while animals are using enrichment techniques and devices. After the table Dr. Duncan discusses that in order to maximize the benefits and minimise the risks of an enrichment technique or device then a zookeeper and the veterinary staff should create effective enrichment for the animals by discussing enrichment applications and possible health risks that might occur. In conclusion Dr. Duncan again states that environmental enrichment provides physiological and behavioral well-being and must continue to be demonstrated to the public as well as animal welfare organizations in order to increase understanding and appreciation of zoo animals being that they are ambassadors to wild creatures.
Overall I thought this paper was easy to understand despite the fourth and fifth conclusions which used quite a lot of veterinary medical terminology. The table that was included was incredibly easy to understand. Based on primarily on the table I now have a better understanding of the potential risks that are involved with environmental enrichment
South Carolina Aquarium Blog Posts Abstract
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina Aquarium, 15 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-surf-internet_15.html>.
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina.Aquarium, 19 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-search.html>.
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina Aquarium, 20 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-surf-internet_20.html>.
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina.Aquarium, 21 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-surf-internet_21.html>.
The South Carolina Aquarium is dedicated to being a local, regional, and national model in science-based education as well as collaborating with partners within the field of wildlife conservation. The South Carolina Aquarium’s sea turtle hospital receives injured, unhealthy sea turtles along the Atlantic coast on behalf of the South Carolina Department of Natural Resources. Over the past twelve years of operation the hospital has released over ninety sea turtles back into the wild.
Within the first “Because sea turtles can’t surf the internet” blog post (February 15), food enrichment is discussed, specifically fish popsicles. A video from YouTube is provided to demonstrate the first use of the fish popsicle with a sub-adult loggerhead patient. The February 19 blog post continues to discuss food enrichment, in this case lettuce feeders made from PVC pipes for their juvenile green patients. After a video demonstration of the patients using the feeder, the diet of Kemp's Ridleys and loggerheads are discussed. Since their beaks are made for shelled prey known for foraging, the hospital faculty give these patients live blue crabs (without the claws to lower the risk of injury to the sea turtles) as demonstrated within the video. Within the February 20 blog post mental and physical enrichment are discussed. As a way to provide stimuli within the tanks, the hospital staff placed balls and PVC pipe for the sea turtle patients to enjoy. A video is provided near the end where a green patient is enjoying a massage from a piece of PVC pipe. The last enrichment post from February 21 continues to discuss physical enrichment from PVC pipes to filters and scrub brushes since turtles do having ‘feeling’ on their carapace. In between the photographic and video demonstrations of the PVC pipes, the author also adds that green patients enjoy backscratchers more than loggerheads and Kemp’s Ridleys do, instead they enjoy the waterfalls, from their filter(s), and being scrubbed by a scrub brush.
My main issue with these blog posts was that they were too basic and didn’t contain nearly as much science-related information about enrichment that I hoped. However, I did like the idea of being able to see photographic and video demonstrations of these enrichment devices with the sea turtle patients.
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina.Aquarium, 19 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-search.html>.
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina Aquarium, 20 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-surf-internet_20.html>.
Megan and Kelly. "Because sea turtles can't surf the internet, continued " Weblog post. South Carolina.Aquarium, 21 February 2011. Web. 17 July 2012. <http://seaturtlehospital.blogspot.com/2011/02/because-sea-turtles-cant-surf-internet_21.html>.
The South Carolina Aquarium is dedicated to being a local, regional, and national model in science-based education as well as collaborating with partners within the field of wildlife conservation. The South Carolina Aquarium’s sea turtle hospital receives injured, unhealthy sea turtles along the Atlantic coast on behalf of the South Carolina Department of Natural Resources. Over the past twelve years of operation the hospital has released over ninety sea turtles back into the wild.
Within the first “Because sea turtles can’t surf the internet” blog post (February 15), food enrichment is discussed, specifically fish popsicles. A video from YouTube is provided to demonstrate the first use of the fish popsicle with a sub-adult loggerhead patient. The February 19 blog post continues to discuss food enrichment, in this case lettuce feeders made from PVC pipes for their juvenile green patients. After a video demonstration of the patients using the feeder, the diet of Kemp's Ridleys and loggerheads are discussed. Since their beaks are made for shelled prey known for foraging, the hospital faculty give these patients live blue crabs (without the claws to lower the risk of injury to the sea turtles) as demonstrated within the video. Within the February 20 blog post mental and physical enrichment are discussed. As a way to provide stimuli within the tanks, the hospital staff placed balls and PVC pipe for the sea turtle patients to enjoy. A video is provided near the end where a green patient is enjoying a massage from a piece of PVC pipe. The last enrichment post from February 21 continues to discuss physical enrichment from PVC pipes to filters and scrub brushes since turtles do having ‘feeling’ on their carapace. In between the photographic and video demonstrations of the PVC pipes, the author also adds that green patients enjoy backscratchers more than loggerheads and Kemp’s Ridleys do, instead they enjoy the waterfalls, from their filter(s), and being scrubbed by a scrub brush.
My main issue with these blog posts was that they were too basic and didn’t contain nearly as much science-related information about enrichment that I hoped. However, I did like the idea of being able to see photographic and video demonstrations of these enrichment devices with the sea turtle patients.
New England Aquarium Blog Posts Abstract
McNally, Kerry. "A Great Day for Hide and Seek!" Weblog post. New England Aquarium, 9 May 2009. Web. 5 July 2012. <http://rescue.neaq.org/2009/05/great-day-for-hide-and-seek.php>.
McNally, Kerry. "Enrichment Day in Quincy." Weblog post. New England Aquarium, 13 Apr. 2012. Web. 5 July 2012. <http://rescue.neaq.org/2012/04/enrichment-day-in-quincy.html>.
McNally, Kerry. "Sea Turtle Enrichment: Goose Eats His Greens!" Web log post. New England Aquarium, 25 Apr. 2009. Web. 5 July 2012.<http://rescue.neaq
.org/2009/04/sea-turtle-enrichment-goose-eats-his.php>.
Merigo, Connie. "Sea Turtle Networking in South Carolina." Web log post. New England Aquarium, 5 May 2009. Web. 5 July 2012. <http://rescue
.neaq.org/2009/05/sea-turtle-networking-in-south-carolina.php>.
The New England Aquarium is a credible source since it’s a national leader of marine conservation. Within these blog posts various ways of enrichment with rehabilitated sea turtles are discussed as well as photographs depicting the enrichment device(s) within the sea turtles’ tank(s). Within the “A Great Day for Hide and Seek!” blog post enrichment for two Kemp's ridleys and one green sea turtle are discussed. The enrichment devices included PVC pipe pieces and “little caves for the turtles to hide in. [They] cut Rubbermaid containers in half and weighted them down to keep them on the bottom [of the tanks]” (“A Great Day for Hide and Seek!”). A picture is provided to display a sea turtle hiding within the Rubbermaid cave. Within the “Enrichment Day in Quincy” blog post tactile enrichment is discussed.
The post references the use of a lettuce feeding device (“Sea Turtle Enrichment: Goose Eats His Greens!”) however green sea turtles are the primary species that feeds on vegetation, not loggerheads or Kemp’s ridleys. To compensate, the rehab faculty created a ‘forest’ out of car wash strips. In the end the loggerhead was hesitant, at first, of the forest while the Kemp’s ridley turtles didn’t really notice the enrichment. The post continues with information of other enrichment devices: a large pipe (McNally references that they got this suggestion from the South Carolina Aquarium “Sea Turtle Networking in South Carolina”) for the turtles to stick their heads into as well as a current flowing out of another large pipe. This post includes several photos of the enrichment devices being used by the turtles. Within the “Sea Turtle Enrichment: Goose Eats His Greens!" post environmental enrichment is discussed.
The post begins by discussing types of environmental enrichment; “rocks to rub against, caves to hide in, and vegetation to swim through” (“Sea Turtle Enrichment: Goose Eats His Greens!"). The type of environmental enrichment discussed within this post is a lettuce feeder with a couple of different kinds of lettuce; “The 'lettuce forest,' as we call it, was made to hold the lettuce for [the green sea turtle] to eat as well as give him something to swim or hide in” (“Sea Turtle Enrichment: Goose Eats His Greens!"). Similar to the introduction to most enrichment devices, the turtle was hesitant initially but eventually started to rub its carapace and head on the feeder and eat the lettuce for several hours. Due to the longevity of the device the author claimed that this enrichment is successful. Similar to the previous post discussed pictures were included demonstrating the hesitation and the involvement of the patient with the enrichment device. Within the “Sea Turtle Networking in South Carolina" post the South Carolina Aquarium and their enrichment technique with their rehab sea turtle patients are discussed.
The post begins by referencing a meeting with the Sea Turtle Rescue Program Coordinator and a Senior Biologist at the Aquarium, Kelly Thorvalson, who has a “knowledge of sea turtle biology and rehabilitation is highly regarded and [a contagious] passion for sea turtles” (“Sea Turtle Networking in South Carolina"). The enrichment device that is discussed (and pictured) is a ledge where a turtle patient can rest their head while sleeping. Unfortunately there is not more information on enrichment devices/techniques within this post.
Overall I thought these blog posts were great sources- several pictures of the sea turtles using the various enrichment devices and easy-to-understand wording. These will be a great aid in the future of some enrichment ideas to possibly tweak.
McNally, Kerry. "Enrichment Day in Quincy." Weblog post. New England Aquarium, 13 Apr. 2012. Web. 5 July 2012. <http://rescue.neaq.org/2012/04/enrichment-day-in-quincy.html>.
McNally, Kerry. "Sea Turtle Enrichment: Goose Eats His Greens!" Web log post. New England Aquarium, 25 Apr. 2009. Web. 5 July 2012.<http://rescue.neaq
.org/2009/04/sea-turtle-enrichment-goose-eats-his.php>.
Merigo, Connie. "Sea Turtle Networking in South Carolina." Web log post. New England Aquarium, 5 May 2009. Web. 5 July 2012. <http://rescue
.neaq.org/2009/05/sea-turtle-networking-in-south-carolina.php>.
The New England Aquarium is a credible source since it’s a national leader of marine conservation. Within these blog posts various ways of enrichment with rehabilitated sea turtles are discussed as well as photographs depicting the enrichment device(s) within the sea turtles’ tank(s). Within the “A Great Day for Hide and Seek!” blog post enrichment for two Kemp's ridleys and one green sea turtle are discussed. The enrichment devices included PVC pipe pieces and “little caves for the turtles to hide in. [They] cut Rubbermaid containers in half and weighted them down to keep them on the bottom [of the tanks]” (“A Great Day for Hide and Seek!”). A picture is provided to display a sea turtle hiding within the Rubbermaid cave. Within the “Enrichment Day in Quincy” blog post tactile enrichment is discussed.
The post references the use of a lettuce feeding device (“Sea Turtle Enrichment: Goose Eats His Greens!”) however green sea turtles are the primary species that feeds on vegetation, not loggerheads or Kemp’s ridleys. To compensate, the rehab faculty created a ‘forest’ out of car wash strips. In the end the loggerhead was hesitant, at first, of the forest while the Kemp’s ridley turtles didn’t really notice the enrichment. The post continues with information of other enrichment devices: a large pipe (McNally references that they got this suggestion from the South Carolina Aquarium “Sea Turtle Networking in South Carolina”) for the turtles to stick their heads into as well as a current flowing out of another large pipe. This post includes several photos of the enrichment devices being used by the turtles. Within the “Sea Turtle Enrichment: Goose Eats His Greens!" post environmental enrichment is discussed.
The post begins by discussing types of environmental enrichment; “rocks to rub against, caves to hide in, and vegetation to swim through” (“Sea Turtle Enrichment: Goose Eats His Greens!"). The type of environmental enrichment discussed within this post is a lettuce feeder with a couple of different kinds of lettuce; “The 'lettuce forest,' as we call it, was made to hold the lettuce for [the green sea turtle] to eat as well as give him something to swim or hide in” (“Sea Turtle Enrichment: Goose Eats His Greens!"). Similar to the introduction to most enrichment devices, the turtle was hesitant initially but eventually started to rub its carapace and head on the feeder and eat the lettuce for several hours. Due to the longevity of the device the author claimed that this enrichment is successful. Similar to the previous post discussed pictures were included demonstrating the hesitation and the involvement of the patient with the enrichment device. Within the “Sea Turtle Networking in South Carolina" post the South Carolina Aquarium and their enrichment technique with their rehab sea turtle patients are discussed.
The post begins by referencing a meeting with the Sea Turtle Rescue Program Coordinator and a Senior Biologist at the Aquarium, Kelly Thorvalson, who has a “knowledge of sea turtle biology and rehabilitation is highly regarded and [a contagious] passion for sea turtles” (“Sea Turtle Networking in South Carolina"). The enrichment device that is discussed (and pictured) is a ledge where a turtle patient can rest their head while sleeping. Unfortunately there is not more information on enrichment devices/techniques within this post.
Overall I thought these blog posts were great sources- several pictures of the sea turtles using the various enrichment devices and easy-to-understand wording. These will be a great aid in the future of some enrichment ideas to possibly tweak.
The Biology of Sea Turtles vol. 2 Chapter 16 Abstract
Higgins, Benjamin M. "16 Sea Turtle Husbandry." The Biology of Sea Turtles. Ed. Peter L. Lutz, John A. Musick, and Jeanette Wyneken. Vol. 2. Boca Raton: CRC, 2003. 21-4. Web. 5 July 2012. <http://www.seaturtle.org/PDF/OCR/HigginsBM_2003_InThebiologyofseaturtles
Volume2_p411-440.pdf>.
The Biology of Sea Turtles vol. 2 is a credible source since the three editors all have PhDs, and either associated with numerous marine conservation groups, a university, or has written numerous books. In addition to this Benjamin Higgins is associated with NOAA at the National Marine Fisheries Service Southeast Fisheries Science Center in Texas. While the rest of the textbook focuses on everything between the physiology and history of sea turtles, chapter sixteen was the most relevant to the enrichment project since the chapter’s all about sea turtle husbandry.
Unlike previous sources, Higgins calls enrichment ‘semiwild conditioning’; “Conditioning introduces the turtles to a wild-type environment while keeping them in a controlled and observable space” (21). He then discusses what has been done previous to Kemp Ridley and Loggerhead sea turtles; after approximately two and four weeks these species increased their strength and stamina compared to turtles that were not conditioned. Higgins then describes the dimensions and contents of the nine large pens that were created for a maximum of two-hundred turtles and the procedures that the turtles had to go through before entering the pen(s). The pens “contain[ed] a natural bay bottom, including sand, seagrass, shell rubble, and rock. The bulkhead walls and mesh sides contain a variety of biota, including algae, barnacles, oysters, sponges, soft corals, tunicates, and various species of cnidarians, echinoderms, fish, crabs, shrimp, and mollusks” (21). The next page begins with a diagram of the pens’ dimensions and contents. Higgins then discusses the procedures to properly remove barnacles and algae from the turtles’ carapaces. Feeding is then discussed followed by possible behavior problems that the turtles exhibited: aggressive and passive-aggressive behavior. The juvenile loggerheads tended to be the aggressive turtles. Higgins defines how a turtle typically exhibits aggressive behavior (mostly consisting of chasing, biting (neck and flippers), splashing, and smacks with a flipper). If the aggressive turtle is not removed from the pen then both turtles will become injured as well as agitating the surrounding pens. Passive-aggressive behavior within turtles is exhibited when a turtle is looking for another turtle to ‘torment’. Higgins ends the chapter by discussing various health issues sea turtles can suffer from.
I found this section of Biology of Sea Turtles vol. 2 easy to understand and analyze for this abstract based on the basic definitions and diagrams. I now have a better understand of how to pinpoint aggressive and passive-aggressive behaviors of sea turtles. I believe that this section of this textbook will create a good reference of enrichment ideas.
Volume2_p411-440.pdf>.
The Biology of Sea Turtles vol. 2 is a credible source since the three editors all have PhDs, and either associated with numerous marine conservation groups, a university, or has written numerous books. In addition to this Benjamin Higgins is associated with NOAA at the National Marine Fisheries Service Southeast Fisheries Science Center in Texas. While the rest of the textbook focuses on everything between the physiology and history of sea turtles, chapter sixteen was the most relevant to the enrichment project since the chapter’s all about sea turtle husbandry.
Unlike previous sources, Higgins calls enrichment ‘semiwild conditioning’; “Conditioning introduces the turtles to a wild-type environment while keeping them in a controlled and observable space” (21). He then discusses what has been done previous to Kemp Ridley and Loggerhead sea turtles; after approximately two and four weeks these species increased their strength and stamina compared to turtles that were not conditioned. Higgins then describes the dimensions and contents of the nine large pens that were created for a maximum of two-hundred turtles and the procedures that the turtles had to go through before entering the pen(s). The pens “contain[ed] a natural bay bottom, including sand, seagrass, shell rubble, and rock. The bulkhead walls and mesh sides contain a variety of biota, including algae, barnacles, oysters, sponges, soft corals, tunicates, and various species of cnidarians, echinoderms, fish, crabs, shrimp, and mollusks” (21). The next page begins with a diagram of the pens’ dimensions and contents. Higgins then discusses the procedures to properly remove barnacles and algae from the turtles’ carapaces. Feeding is then discussed followed by possible behavior problems that the turtles exhibited: aggressive and passive-aggressive behavior. The juvenile loggerheads tended to be the aggressive turtles. Higgins defines how a turtle typically exhibits aggressive behavior (mostly consisting of chasing, biting (neck and flippers), splashing, and smacks with a flipper). If the aggressive turtle is not removed from the pen then both turtles will become injured as well as agitating the surrounding pens. Passive-aggressive behavior within turtles is exhibited when a turtle is looking for another turtle to ‘torment’. Higgins ends the chapter by discussing various health issues sea turtles can suffer from.
I found this section of Biology of Sea Turtles vol. 2 easy to understand and analyze for this abstract based on the basic definitions and diagrams. I now have a better understand of how to pinpoint aggressive and passive-aggressive behaviors of sea turtles. I believe that this section of this textbook will create a good reference of enrichment ideas.
"Experimental Evaluation of Environmental Enrichment of Sea Turtles." Abstract
Therrien, C. L. ; Gaster, L. ; Cunningham-Smith, P. ; Manire, C. A. "Experimental Evaluation of Environmental Enrichment of Sea Turtles." Zoo Biology 2007, v. 26, no. 5, p.407-416.<http://florida.academia.edu/PetraCunninghamSmith/Papers/872703/Experimental_evaluation_of_environmental_enrichment_of_sea_turtles> (10 pages).
This journal article is sponsored by the Mote Marine Laboratory and Aquarium, located in Florida, who is a leader in marine research since 1955- making this a credible source. The introduction begins by referencing psychological and physical benefits of enrichment for captive animals, lists facts and difficulties that go in hand with studying enrichment with sea turtles, such as the need for documentation on “[b]ehaviors relating to hunting, foraging, or socializing” (2). Despite the lack of previous information and studies, the Mote Marine Aquarium created “an environmental enrichment program [which] was needed for four display turtles to offset the effects of a somewhat sterile single species environment. The program use[d] enrichment devices that would provide opportunities to forage for food, to increase exploratory swimming and use of available habitat, to stimulate tactile sensitivity, and to reduce stereotypic swimming. To fill the aforementioned information gap, the following experiment was designed to determine the effects of enrichment on the sea turtles, including those of
a special needs (blind) turtle” (2).
On page three a table displays the descriptions of the four sea turtles that were included within the study. Within the methods section, tank dimensions, temperature, salinity, and chlorine levels are discussed. Then descriptions and various wanted behaviors were explained with each enrichment device as well as pictures (of each device). The authors then clarify how they adjusted the various enrichment devices for the blind sea turtle. In order to collect data the researchers created eight categories of behaviors and used a timer for when a turtle exhibited a said behavior (4). In order to compare behaviors with vs. without enrichment, statistics were used in order to create a bar graph based on table 3 (5). In the end, each of the turtles were affected differently with each enrichment device due to individual behavioral differences. However, “[a]ll four turtles exhibited highly significant increases in Random Swimming and Focused Behavior and highly significant decreases in Pattern Swimming and Resting (Table 4)” (4); in other words, “ environmental enrichment can be effective in increasing exploratory and novel behaviors in captive marine reptiles” (5). Due to the success of this 2004 study, the turtles improved their psychological and physiological health through the use of these various devices.
I found this journal article easy to understand and analyze for this abstract based on basic wording and an easy-to-understand tables. I now have a better understanding of enrichment for rehabilitating sea turtles. I believe that this will aid me in the future as an idea of how to collect data as well as how to analyze it afterwards. This study was also helpful to me since it demonstrated that enrichment can be made effective for handicapped sea turtles, which is important since the PPG Aquarium has a green sea turtle with dysfunctional front flippers.
This journal article is sponsored by the Mote Marine Laboratory and Aquarium, located in Florida, who is a leader in marine research since 1955- making this a credible source. The introduction begins by referencing psychological and physical benefits of enrichment for captive animals, lists facts and difficulties that go in hand with studying enrichment with sea turtles, such as the need for documentation on “[b]ehaviors relating to hunting, foraging, or socializing” (2). Despite the lack of previous information and studies, the Mote Marine Aquarium created “an environmental enrichment program [which] was needed for four display turtles to offset the effects of a somewhat sterile single species environment. The program use[d] enrichment devices that would provide opportunities to forage for food, to increase exploratory swimming and use of available habitat, to stimulate tactile sensitivity, and to reduce stereotypic swimming. To fill the aforementioned information gap, the following experiment was designed to determine the effects of enrichment on the sea turtles, including those of
a special needs (blind) turtle” (2).
On page three a table displays the descriptions of the four sea turtles that were included within the study. Within the methods section, tank dimensions, temperature, salinity, and chlorine levels are discussed. Then descriptions and various wanted behaviors were explained with each enrichment device as well as pictures (of each device). The authors then clarify how they adjusted the various enrichment devices for the blind sea turtle. In order to collect data the researchers created eight categories of behaviors and used a timer for when a turtle exhibited a said behavior (4). In order to compare behaviors with vs. without enrichment, statistics were used in order to create a bar graph based on table 3 (5). In the end, each of the turtles were affected differently with each enrichment device due to individual behavioral differences. However, “[a]ll four turtles exhibited highly significant increases in Random Swimming and Focused Behavior and highly significant decreases in Pattern Swimming and Resting (Table 4)” (4); in other words, “ environmental enrichment can be effective in increasing exploratory and novel behaviors in captive marine reptiles” (5). Due to the success of this 2004 study, the turtles improved their psychological and physiological health through the use of these various devices.
I found this journal article easy to understand and analyze for this abstract based on basic wording and an easy-to-understand tables. I now have a better understanding of enrichment for rehabilitating sea turtles. I believe that this will aid me in the future as an idea of how to collect data as well as how to analyze it afterwards. This study was also helpful to me since it demonstrated that enrichment can be made effective for handicapped sea turtles, which is important since the PPG Aquarium has a green sea turtle with dysfunctional front flippers.
"Marine Turtle Trauma Response Procedures: A Husbandry Manual" Abstract
Bluvias, Jessie E., and Dr. Karen L. Eckert ed. "MARINE TURTLE TRAUMA RESPONSE PROCEDURES: A HUSBANDRY MANUAL." Duke University, May 2008. Web. 19 June 2012. <http://dukespace.lib.duke.edu/dspace/bitstream/handle/1016/464/MP_jeb30_a_05206.08.pdf;jsessionid=BF8A76D31C3904D77FB999538E9A
8225?sequence=1>. p. 1-8, 34-5
This document is Bluvias’ Master’s project for a Master degree in Environmental Management from Duke University (wrote with guidance of Dr. Eckert, the author’s adviser). Being that Bluvias plans to publish this in several languages and distributed by the Wider Caribbean Sea Turtle Conservation Network (WIDECAST) “to under-resourced Caribbean conservationists, managers, and facilities staff in the Wider Caribbean Region [( which includes the United States, Mexico, Central America, the northern countries of South America, and all of the Caribbean islands, where six out of seven species of sea turtles inhabit (1))]” (1) this document is a credible source. Throughout this document the author describes to the audience several methods “to encourage and facilitate the best professional care of sick and injured sea turtles during their rehabilitation” (2) by examining ways in which scientists have gathered information for husbandry protocols (2-3). Bluvias begins by informing the audience of basic background of sea turtles: where they live, common threats that sea turtles undergo, as well as husbandry protocols that are practiced by the Association of Zoos and Aquariums (AZA) and the UN Regional Seas Convention in the Wider Caribbean Region. Within the introduction, another goal of the author becomes clear- to help make standard guidelines for rehabilitating sea turtles since “[q]uestions range from wondering about sample diets, tank sizes, and water quality standards to enrichment ideas (Archives of [email protected], 2007). The need for standard guidelines and criteria is evident in the questions sent...and the thought of designing a guide to answer these common questions has been around for several years” (2). The need of guidelines for husbandry protocol of sea turtles is at large since it relates “to the care of federally and internationally protected species” (2). By examining these sources, it is evident that Bluvias intended “to create a manual to help standardize husbandry protocols for AZA member institutions, implement federal sea turtle recovery plan tasks, and offer guidance to Caribbean-based caregivers to increase the survival chances for sick and injured sea turtles” (4).
Within the methods section, the author also gained his information by interning at three sea turtle facilities where he participated in daily husbandry procedures (“including food preparation, dispensing medicine, feeding, cleaning tanks, water quality testing, transporting, holding for diagnostic procedures, and addressing wounds” (5)) and collaborating with various sea turtle experts, veterinarians, and professional sea turtle caregivers. Bluvias then describes the table of contents within his manual and a general overview of it (6). The main challenges that he encountered during the creation of his manual was that each of the three facilities that he interned at had different “funding, staff numbers, skills (which varied from untrained volunteers to trained technicians), mandates, and husbandry protocols such as physical plant and diet” (7). Another issue that he faced was intending the manual for third-world countries. Bluvias overcame both of these by “offering basic protocols, a variety of options, and recommendations, in addition to researching and presenting high and low-tech solutions for the husbandry objectives” (8).
Personally, I found this article easy to understand and analyze for this abstract. The table that is displayed (3) was somewhat confusing understand. I now recognize the difficulties that sea turtles encounter and the issues that was making rehabilitating sea turtles an intricate process for sea turtle handlers and veterinarians alike. I believe that this article (and the manual) will help me greatly in the future as a way to understand basic husbandry and rehabilitation processes of sea turtles.
________________________________________
Within pages thirty-four and thirty-five Bluvias discusses enrichment techniques for rehabilitated sea turtles. He defines enrichment as a way to “provide behavioral choices by allowing turtles to use the available space, reduces stereotypical swimming that can result in injury (such as calluses from rubbing the sides of the tank), and encourages species-specific activity related to exploration, foraging, and tactile stimulation (Cunningham-Smith et al., undated; Therrien et al., 2007). The promotion of natural behaviors aids in the rehabilitation process by stimulating appetite, building strength, and encouraging alertness” (34). He then divides enrichment items into two categories: ‘food-based enrichment’ and ‘non-food-based enrichment’, both are necessary to rehabilitate sea turtles. The author gives the audience detailed instructions of how to properly use and clean these various items. Bluvias defines food-based enrichment as a way to “best stimulate appetite, curiosity, and movement” (34) of sea turtles. Some examples of food-based enrichment items include: live food, ice blocks, feeding mats, and feeding tubes. He then defines non-food-based enrichment “for tactile stimulation and to promote natural exploration. These items can be left in tanks during hours of supervision. Items should never be shared between tanks unless they are sterilized between uses” (35). Some items include rocks, waterfalls, hiding places, and back scratchers. Bluvias concludes the enrichment section of his manual with a concerns and warnings section; “Be mindful when designing enrichment items: keep the design simple and as natural as possible. Turtles may associate certain materials and objects with something they would find in the wild like parking cones, buoys, or tires. This association encourages exploration and alertness. Enrichment items are also meant for turtles that are in better health and should not be used for debilitated turtles, unless live food is given to stimulate appetite” (35).
Again, I found this section of Bluvias’ manual easy to understand and analyze for this abstract based on basic definitions, pictures, and easy-to-understand instructions of each enrichment item. I now have a better understanding of various types and items used for enrichment purposes with rehabilitating sea turtles. I believe that this section of the manual will aid me in the future as a way basis of enrichment ideas for the sea turtles.
8225?sequence=1>. p. 1-8, 34-5
This document is Bluvias’ Master’s project for a Master degree in Environmental Management from Duke University (wrote with guidance of Dr. Eckert, the author’s adviser). Being that Bluvias plans to publish this in several languages and distributed by the Wider Caribbean Sea Turtle Conservation Network (WIDECAST) “to under-resourced Caribbean conservationists, managers, and facilities staff in the Wider Caribbean Region [( which includes the United States, Mexico, Central America, the northern countries of South America, and all of the Caribbean islands, where six out of seven species of sea turtles inhabit (1))]” (1) this document is a credible source. Throughout this document the author describes to the audience several methods “to encourage and facilitate the best professional care of sick and injured sea turtles during their rehabilitation” (2) by examining ways in which scientists have gathered information for husbandry protocols (2-3). Bluvias begins by informing the audience of basic background of sea turtles: where they live, common threats that sea turtles undergo, as well as husbandry protocols that are practiced by the Association of Zoos and Aquariums (AZA) and the UN Regional Seas Convention in the Wider Caribbean Region. Within the introduction, another goal of the author becomes clear- to help make standard guidelines for rehabilitating sea turtles since “[q]uestions range from wondering about sample diets, tank sizes, and water quality standards to enrichment ideas (Archives of [email protected], 2007). The need for standard guidelines and criteria is evident in the questions sent...and the thought of designing a guide to answer these common questions has been around for several years” (2). The need of guidelines for husbandry protocol of sea turtles is at large since it relates “to the care of federally and internationally protected species” (2). By examining these sources, it is evident that Bluvias intended “to create a manual to help standardize husbandry protocols for AZA member institutions, implement federal sea turtle recovery plan tasks, and offer guidance to Caribbean-based caregivers to increase the survival chances for sick and injured sea turtles” (4).
Within the methods section, the author also gained his information by interning at three sea turtle facilities where he participated in daily husbandry procedures (“including food preparation, dispensing medicine, feeding, cleaning tanks, water quality testing, transporting, holding for diagnostic procedures, and addressing wounds” (5)) and collaborating with various sea turtle experts, veterinarians, and professional sea turtle caregivers. Bluvias then describes the table of contents within his manual and a general overview of it (6). The main challenges that he encountered during the creation of his manual was that each of the three facilities that he interned at had different “funding, staff numbers, skills (which varied from untrained volunteers to trained technicians), mandates, and husbandry protocols such as physical plant and diet” (7). Another issue that he faced was intending the manual for third-world countries. Bluvias overcame both of these by “offering basic protocols, a variety of options, and recommendations, in addition to researching and presenting high and low-tech solutions for the husbandry objectives” (8).
Personally, I found this article easy to understand and analyze for this abstract. The table that is displayed (3) was somewhat confusing understand. I now recognize the difficulties that sea turtles encounter and the issues that was making rehabilitating sea turtles an intricate process for sea turtle handlers and veterinarians alike. I believe that this article (and the manual) will help me greatly in the future as a way to understand basic husbandry and rehabilitation processes of sea turtles.
________________________________________
Within pages thirty-four and thirty-five Bluvias discusses enrichment techniques for rehabilitated sea turtles. He defines enrichment as a way to “provide behavioral choices by allowing turtles to use the available space, reduces stereotypical swimming that can result in injury (such as calluses from rubbing the sides of the tank), and encourages species-specific activity related to exploration, foraging, and tactile stimulation (Cunningham-Smith et al., undated; Therrien et al., 2007). The promotion of natural behaviors aids in the rehabilitation process by stimulating appetite, building strength, and encouraging alertness” (34). He then divides enrichment items into two categories: ‘food-based enrichment’ and ‘non-food-based enrichment’, both are necessary to rehabilitate sea turtles. The author gives the audience detailed instructions of how to properly use and clean these various items. Bluvias defines food-based enrichment as a way to “best stimulate appetite, curiosity, and movement” (34) of sea turtles. Some examples of food-based enrichment items include: live food, ice blocks, feeding mats, and feeding tubes. He then defines non-food-based enrichment “for tactile stimulation and to promote natural exploration. These items can be left in tanks during hours of supervision. Items should never be shared between tanks unless they are sterilized between uses” (35). Some items include rocks, waterfalls, hiding places, and back scratchers. Bluvias concludes the enrichment section of his manual with a concerns and warnings section; “Be mindful when designing enrichment items: keep the design simple and as natural as possible. Turtles may associate certain materials and objects with something they would find in the wild like parking cones, buoys, or tires. This association encourages exploration and alertness. Enrichment items are also meant for turtles that are in better health and should not be used for debilitated turtles, unless live food is given to stimulate appetite” (35).
Again, I found this section of Bluvias’ manual easy to understand and analyze for this abstract based on basic definitions, pictures, and easy-to-understand instructions of each enrichment item. I now have a better understanding of various types and items used for enrichment purposes with rehabilitating sea turtles. I believe that this section of the manual will aid me in the future as a way basis of enrichment ideas for the sea turtles.
